Human Pancreatic Microvascular Endothelial Cells
Cat.No.: CSC-C4366X
Species: Human
Source: Pancreas
Cell Type: Endothelial Cell; Microvascular Cell
- Specification
- Background
- Scientific Data
- Q & A
- Customer Review
Human Pancreatic Microvascular Endothelial Cells (HPMECs) are primary endothelial cells derived from human pancreatic microvasculature. They offer an in vitro model system that is physiologically relevant for studying pancreatic vascular biology and endothelial cell function. These cells contribute to homeostasis of pancreatic tissue and regulate nutrient delivery, barrier function and paracrine support of the pancreatic microenvironment.
HPMECs have a classic cobblestone endothelial cell morphology and under normal culture conditions they exhibit tight junctions and grow to confluency as a monolayer. They are positive for endothelial cell markers including CD31 (PECAM-1), VE-cadherin, von Willebrand factor (vWF) and endothelial nitric oxide synthase (eNOS). HPMECs are responsive to angiogenic and inflammatory cues and possess regulated permeability and nitric oxide-mediated responses.
HPMECs have been utilized in studies to model pancreatic microcirculation, angiogenesis, endothelial cell dysfunction and responses to metabolic and inflammatory stimuli involved in diseases such as diabetes mellitus and pancreatitis. These models are particularly useful to study endothelial-islet cell interactions, vascular remodeling, pancreatic inflammation and injury. HPMECs can also be used to screen vascular-targeted and/or anti-inflammatory therapies.
PDAC EVs are Readily Imported by Endothelial Cells in vitro
Pancreatic ductal adenocarcinoma (PDAC) is hypovascular with collapsed vessels that limit immunosurveillance and promote metastasis; antiangiogenic therapies have failed. Finan et al. investigated whether the RNA-binding protein HuR in cancer cells reshapes the tumor microenvironment by regulating extracellular vesicle (EV) cargo that signals to endothelial cells.
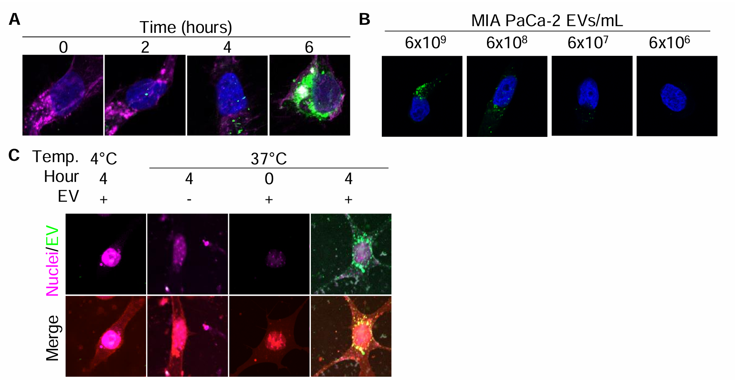
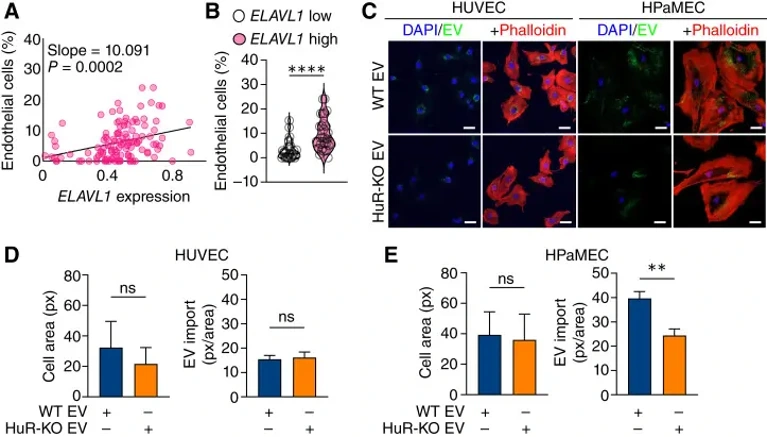
To assess whether endothelial cells import human PDAC EVs, they optimized in vitro EV import studies using the lipophilic dye PKH67. Endothelial cells treated with PKH67-labeled EVs showed significant EV import by 4 hours, after which the lipid signal became diffuse as lipids were recycled (Fig. 1A). EV import was concentration-dependent, so subsequent studies used the concentration released by PDAC cells over 48 hours (~300 EVs/cell; Fig. 1B). Control experiments confirmed that PKH67 alone at 37°C or PKH67-labeled EVs at 4°C produced no signal, validating active EV import monitoring (Fig. 1C). Using this approach, they observed that both HUVECs and HPaMECs imported PDAC EVs (Fig. 2C-E). Notably, HPaMECs-but not HUVECs-imported significantly fewer HuR-KO EVs than WT EVs, consistent with reports that EV import is a regulated process involving tissue- or cell-specific receptor-ligand interactions. These findings validate previous reports that endothelial cells import PDAC EVs in vitro.
Ask a Question
Write your own review