Physicochemical Characterization Assays
Creative Bioarray provides Physicochemical Characterization Assays to help customers accurately evaluate compounds' physicochemical properties such as solubility and stability, Reducing drug development difficulties.
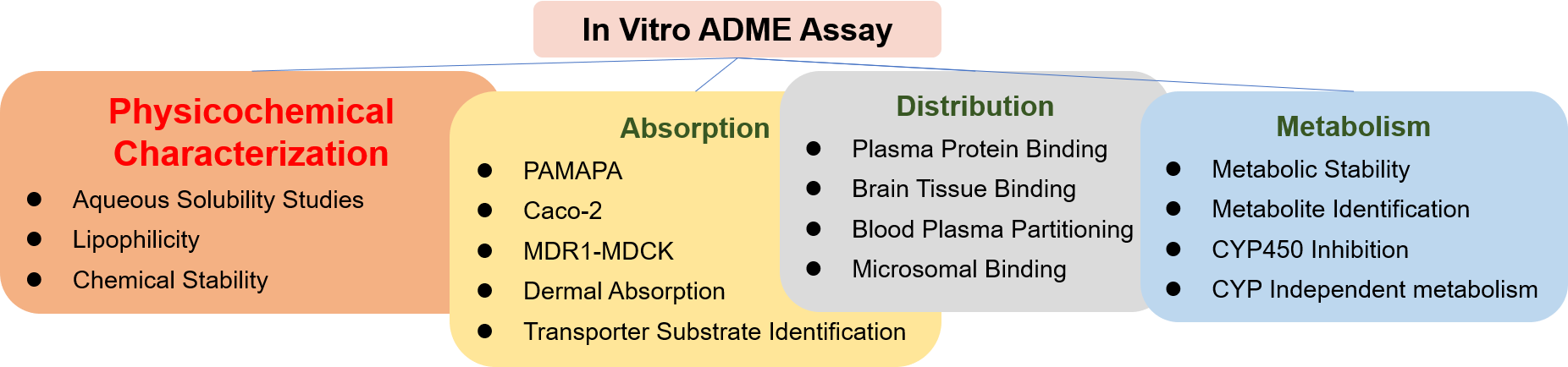
The Importance of Physicochemical Characterization Assays
- Knowledge of compounds' physicochemical properties such as solubility and stability is essential for rationalizing drug formulations, reducing difficulties in drug development, and controlling the effects of drug action.
- The most important goal in drug delivery is to bring the drug concentration to a specific level and maintain it at that level for a specific duration of time. Stability and solubility are two fundamental physicochemical properties that must be considered when designing a successful drug formulation.
- The physicochemical properties of the drug in both solution and solid state play a critical role in drug formulation. Solid-state formulations are generally favored over liquid formulations because they are more chemically stable, easier to process, and easier to deliver. If the substance is solid, it must dissolve before it can be therapeutically active, and once in solution, it must be both soluble and chemically stable.
Our Capabilities
Low solubility can impair the formation of in vitro DMPK or biological data quality, which can be a significant stumbling block in the drug discovery and development process. It may lead to problems in generating suitable formulations for in vivo pharmacokinetic studies. Unknown solubility may lead to problems in the absorption of the drug after oral administration. If the drug solubility is known, then the correct and reliable compound concentration will be available.
- Kinetic solubility assay (The most common method)
- Thermodynamic solubility assay
- Lipophilicity Assays
Lipophilicity and pKa are important factors in permeability and solubility, and they're frequently tested during drug development.
The assessment of lipophilicity is fundamental to the understanding of molecular properties. In a drug development context, it's also vital to consider about ionization of molecules in the aqueous phase, especially at pH 7.4. Therefore, the distribution coefficient (Log D) is the preferred descriptor of lipophilicity.
The distribution of a molecule between a lipid or nonpolar environment and an aqueous or polar environment is affected by lipophilicity. The equilibrium distribution of a chemical between water and octanol is commonly quantified as Log P. Lower aqueous solubility and higher membrane permeability are associated with compounds with a higher Log P.
- Log D
- Log P
- pKa
pKa influences the ionization of molecules in an aqueous solution; it is measured by the change of ionization with pH. Basic compounds with pKa of 9 have an even distribution of protonated and neutral molecules at pH=9. The lower the pH, the higher the protonation and the more neutral species. Acidic compounds with pKa of 4 have an equal distribution of deprotonated and neutral molecules at pH=4, with more neutral species at low pH and more deprotonated species at high pH.
Chemical stability of the drug includes reactions such as hydrolysis, dehydration, oxidation, photochemical degradation, or reaction with excipients. The constant presence of water and oxygen in our environment means that exposure to moisture or oxygen can affect chemical stability. Oral drugs are also exposed to an acidic environment as they pass through the gastrointestinal tract, compromising chemical stability. Chemical stability is essential not only because a sufficient amount of the drug is needed at the time of administration for therapeutic purposes but also because chemical degradation products may adversely affect the properties of the formulated product and may even be toxic.
- Stability in different pH buffers
- Stability in simulated gastric fluid (SGF)
- Stability in simulated intestinal fluid (SIF)
Quotation and ordering
If you have any special needs or questions regarding our services, please feel free to contact us. We look forward to cooperating with you in the future.
Reference
- Barich, Dewey H., Eric J. Munson, and Mark T. Zell. "Physicochemical properties, formulation, and drug delivery." Drug Delivery: Principles and Applications (2005): 57-71.
Explore Other Options