Microsomal Stability Assay
- Service Details
- Features
- FAQ
- Explore Other Options
The microsomal stability assay is a popular in vitro ADME test that measures how quickly a molecule is digested by liver drug-metabolizing enzymes. This test offers an effective method of evaluating metabolic stability without the complexity of whole-cell systems by using hepatic microsomes enriched in phase I and phase II enzymes, such as cytochrome P450 (CYPs) and UDP-glucuronosyltransferases (UGTs).
During the early stages of drug discovery, microsomal stability data are frequently utilized to rank compounds according to metabolic liability, estimate intrinsic clearance (CLint), and direct medicinal chemistry optimization.
Why microsomal stability matters in drug development
Poor metabolic stability is one of the leading causes of attrition in drug development. Early identification of rapidly cleared compounds helps teams:
- Prioritize stable leads before costly in vivo studies
- Optimize chemical structures to improve half-life and exposure
- Predict hepatic clearance and dosing challenges
- Reduce late-stage ADME-related failures
Microsomal stability assays offer a fast and scalable solution for early metabolic risk assessment.
Our Microsomal Stability Assay Service
Workflow
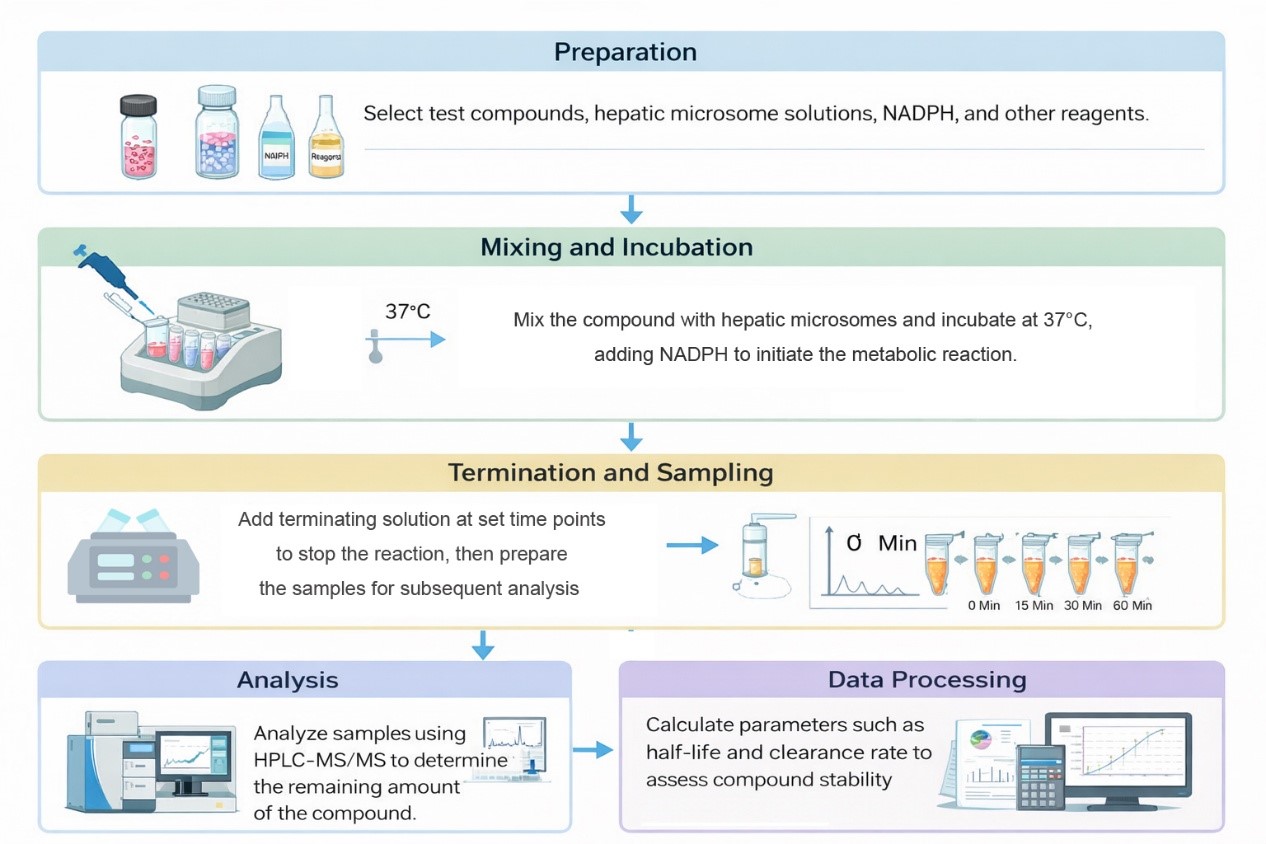
| Microsomal Stability Assay | Details |
| Species | Human, rat, mouse, (other available) |
| Test Compound Concentration | 1 µM (different concentrations available on request) |
| Microsomes Concentration | 0.5 mg/mL (different concentrations available on request) |
| Time Points | 0, 5, 15, 30, 45 and 60 minutes |
| Replicates | 2 |
| Cofactor | 1 mM NADPH (other cofactors available on request) |
| Compound Requirements | 50 µL of 10 mM DMSO solution or 1 mg of solid material |
| Negative Control |
Vehicle (0.1% DMSO) Heat inactivated microsomes |
| Positive Controls |
Verapamil (rapid clearance) Diazepam (low clearance) |
| Analysis Method | HPLC-MS/MS |
| Data Delivery | Apparent intrinsic clearance (CLint, app), half-life (t1/2), parent remaining (%) including standard error of mean (SEM) |
More metabolic stability assays:
Creative Bioarray's microsomal stability assay can be further extended to metabolite profiling and give out more information about the metabolism of your compound.
Conduct metabolic studies using complete human hepatocyte models to provide cellular-level metabolic information, aiding in the prediction of a drug's clearance rate in vivo.
Perform metabolic stability analysis using liver S9 fractions, combining phase I and II metabolic evaluations for a more comprehensive understanding of the compound's metabolic pathways.
Why Choose Our Microsomal Stability Assay?
Broad Enzyme Coverage
Liver microsomes contain key phase I and II metabolic enzymes, enabling comprehensive assessment of metabolic pathways.
Multi-Species Capability
Human, rat, mouse, and other species microsomes available to support interspecies comparison and translational studies.

Flexible Study Design
Custom compound concentrations, incubation times, and cofactors available upon request.
Low Material Requirement
Minimal compound input—ideal for early discovery programs with limited material.

Expert ADME Support
Our experienced scientists assist with data interpretation and study design optimization.
FAQ
1. What is metabolic stability analysis?
Metabolic stability analysis evaluates the degradation rate of compounds in biological samples (e.g., liver microsomes, hepatocytes, S9 fractions). This analysis provides insights into the in vivo metabolic stability, half-life, and clearance rate of compounds, which are crucial for predicting their pharmacokinetic properties during drug development.
2. What are the advantages of using liver microsomes for drug metabolism research?
Liver microsomes from hepatocyte endoplasmic reticulum contain many CYP450 enzymes that drive drug metabolism. Advantages include:
- Rich in Metabolic Enzymes: Contains abundant CYP450 enzyme systems to simulate the main drug metabolism process.
- Simple Operation: Easy to prepare with controllable incubation conditions, suitable for high-throughput screening.
- Low Cost: More cost-effective to prepare and use than complete hepatocytes.
- Predicting In Vivo Clearance Rate: Measuring liver microsomal metabolism helps scientists estimate actual drug clearance rates to support pharmacokinetic analysis.
3. What are the differences between hepatocyte stability and microsomal stability?
- Enzyme System Differences: Hepatocytes possess enzymes aldehyde oxidase (AO) and xanthine oxidase (XO) that help metabolism but these enzymes exist in lower amounts or lack entirely in microsomal preparations.
- Structural Differences: Hepatocytes have intact structures including cell membranes, whereas microsomes are non-structured enzymatic sources, leading to differences in permeability and uptake/excretion processes.
- Transporter Effects: Hepatocytes express various drug transporters (e.g., OATP, BCRP) that affect uptake and excretion, impacting metabolic rates, whereas microsomes do not contain these transporters.
4. Why can microsomal CLint data predict in vivo clearance rate?
Microsomal intrinsic clearance (CLint) indicates the rate at which a compound is metabolized by enzymes in liver microsomes. It forms the basis for predicting in vivo clearance as the liver is the main metabolic organ. CLint reflects a drug's potential hepatic clearance capacity and can be extrapolated to whole-body clearance through proper modeling and scaling, crucial for drug dosing and optimization.
5. How is microsomal stability data interpreted?
Key parameters include:
- Half-life (t1/2): Time for drug concentration to decline by half. A longer half-life indicates slower metabolism and better stability.
- Intrinsic Clearance (CLint): Volume of drug metabolized per unit time. Lower CLint indicates slower metabolism and lower in vivo clearance.
- Metabolite Formation: Identifying metabolites helps understand primary metabolic pathways and aids in drug structure optimization.
Need Reliable Microsomal Stability Data?
Partner with our ADME specialists to accelerate your drug development program.
Explore Other Options