CAR-T Preclinical Characterization in vivo
Chimeric antigen receptors (CARs) are receptor proteins that are engineered to give T cells new abilities to recognize specific targets. The receptors are chimeric because they combine both antigen-binding and T-cell activating functions into a single receptor. The premise of CAR-T immunotherapy is to modify T cells to recognize cancer cells in order to more effectively target and destroy them.
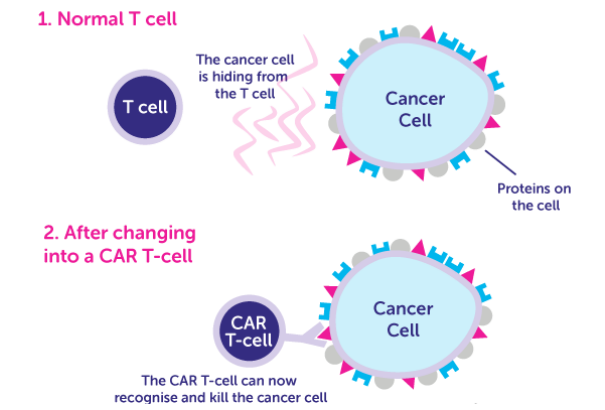
In order to apply CAR-T therapy in more conditions effectively and efficiently, preclinical assays on animal models are necessary for approval of clinical trials based on CAR-T therapy. The in vivo assays of CAR-T therapy mainly focus on characteristics, working mechanism, applicable conditions, suitable population, pathways, and target sites of CAR-T medical products.
Pharmacological, as well as Pharmacokinetic, Evaluation are Essential for CAR-T Therapies (including but not limited to)
- Target expression detection includes target binding sites and target expressing sites
- Functional analysis includes proliferation assay, cytokine secretion detection, targeting antigen distribution assay, and affinity analysis
- Anti-tumor efficacy includes tumor-killing ability detection
- In vivo Biodistribution analysis in vivo includes CAR-T exposure level detection by time, retaining duration assay, and elimination assay by time
Additionally, Safety Assessment Needs to be Carefully Monitored (including but not limited to)
- CAR-T immune response analysis involves primary dose of CAR-T cells and cytotoxicity reaction
- CAR-T cytotoxicity effect detection involves neurotoxicity, immunotoxicity, and etc.
- Risk analysis of insertional mutation involves immortalized proliferation or tumorigenicity risk
- Long-term cytotoxicity and tumorigenicity assay involve insertion site analysis
- Cytokine release syndrome (CRS) analysis involves neurotoxicity, encephaledema, and etc.
- Safety analysis of virus vector involves type, biodistribution, proliferating level etc. of virus
- Risk of choice for model animals involves normal or disease-model animals
All of the in vivo preclinical characterization share the same principle: integration of pharmacological, pharmacokinetic, and safety analysis to establish the most efficient and cost-saving experiment for CAR-T medical products and CAR-T cells.
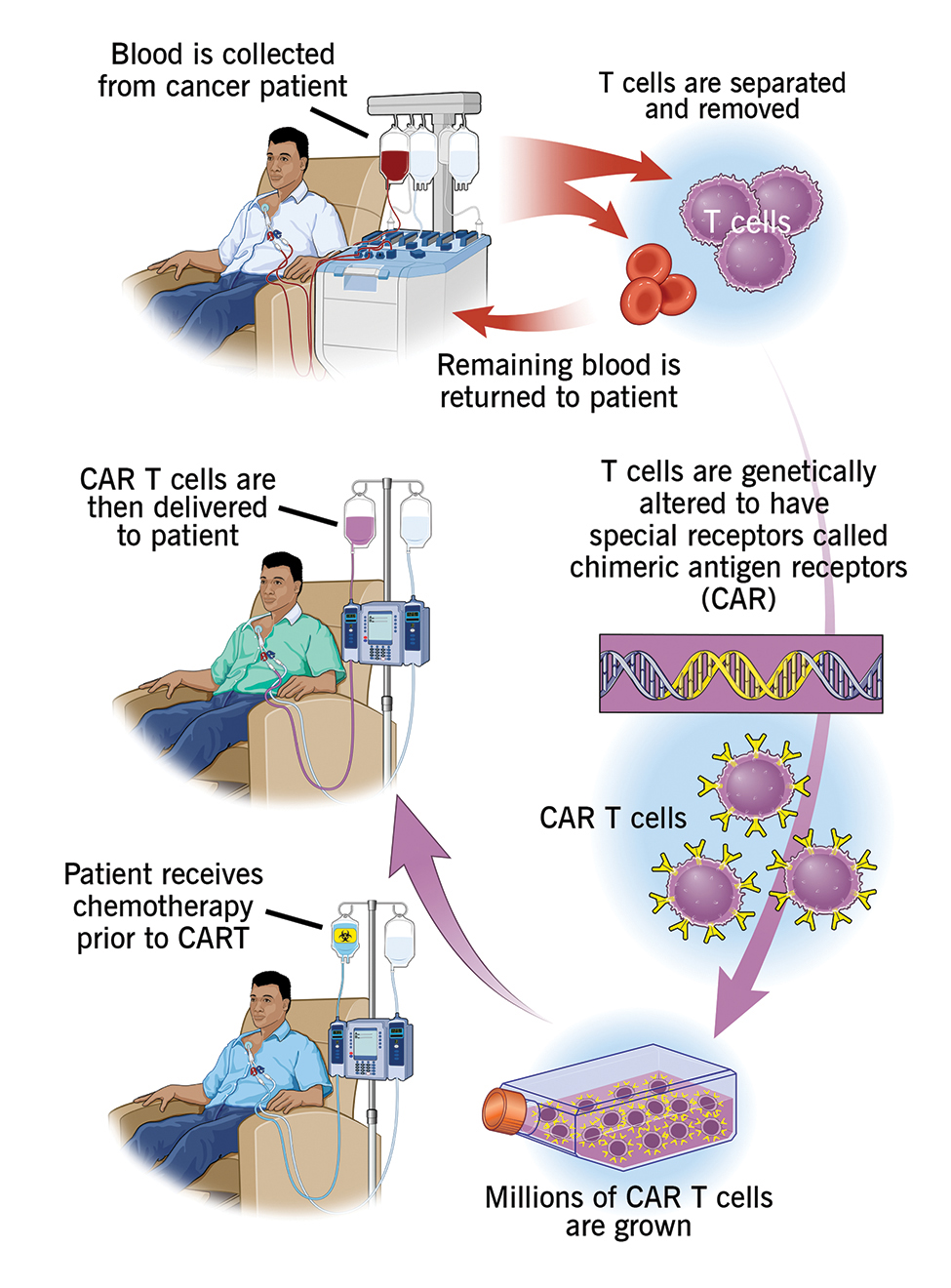
Creative Bioarray is an experienced and outstanding provider of CAR-T Preclinical Characterization. We are dedicated to providing detailed data and comprehensive service for your scientific research, and we are pleased to use our advanced platform and professional experience to satisfy your demand for you.

If you have any special need in CAR-T Preclinical Characterization, do not hesitate to contact us for this special service. Please let us know what you need and we will accommodate you. We are looking forward to working with you in the future.
Explore Other Options