How Are Biomarkers Validated in Drug Development?
What Is a Biomarker? A biomarker is any measurable indicator of biological processes occurring within the human body. It could be a protein found in blood, a specific gene, or even the results from a PET or MRI image. Biomarkers are heavily utilized in drug discovery and can help researchers gain a better understanding of disease pathology, track patients' response to treatment, and ultimately make more informed decisions during clinical trials.
But a biomarker alone is meaningless without verification. False signals and invalid results could be sent your way if your data isn't validated properly. That is why regulators such as the FDA and EMA are focusing more efforts on fit-for-purpose validation and defining context of use (COU). Ultimately, proper biomarker validation will lead to faster drug discovery, more robust clinical trials, and better precision medicine results.
The Significance of Biomarker Validation
If a biomarker isn't properly validated, it could do more harm than good. You could be sent false signals leading to false conclusions and ultimately failed clinical trials. All of this costs time and money that could have been spent elsewhere.
On the other hand, clinical trials can go much smoother with well validated biomarkers. They can help researchers better select patients, reduce trial sizes, and increase chances for success. From a regulator's standpoint, biomarkers can also help decide if a drug should be approved, what it should be labelled as, and dosage amount.
There are many real-world examples where validated biomarkers are making an impact. Oncologists are using protein-based biomarkers to make decisions on patient immunotherapy. In neurology, doctors are using imaging biomarkers to track the progression of diseases like Alzheimer's. These are just a few examples of how validation really gives biomarkers value.
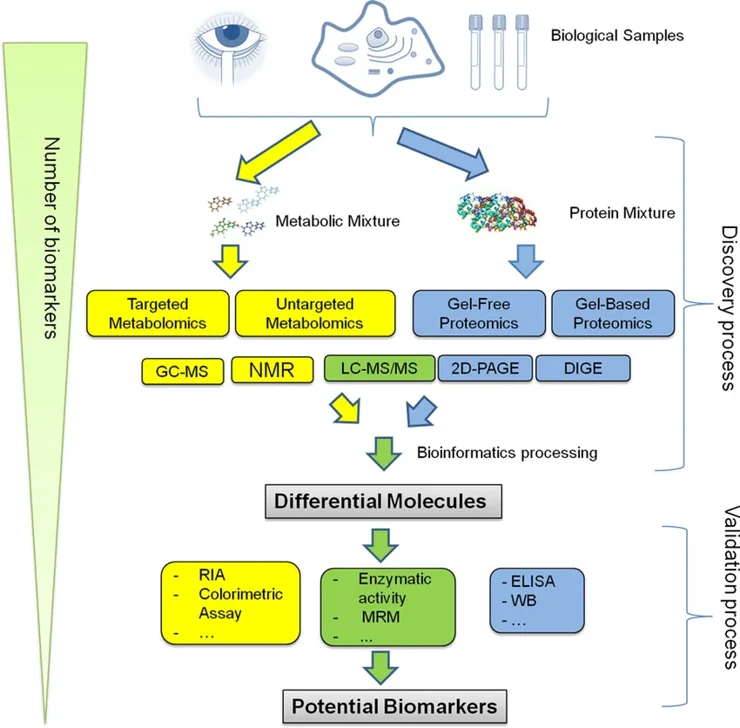
Validation Approaches of Biomarkers
Different types of Biomarkers require slightly different approaches to validation. Let's discuss these major types and what you need to consider when validating each type. The more you know about the differences between these biomarker types upfront, the easier it will be to select appropriate validation methods and save thousands (sometimes millions) of dollars in drug development costs down the road.
Molecular Biomarkers
The most commonly used type of biomarkers are molecular biomarkers. These include proteins, mutations in DNA, messenger RNA (mRNA) expression levels, and metabolites. These biomarkers are typically collected from a blood sample, tissue sample, or other biospecimen.
Common detection technologies include:
- Ligand-binding assays (LBA) (e.g., ELISA) for protein quantification
- PCR/qPCR for gene expression and mutation detection
- Next-generation sequencing (NGS) for genomic profiling
- LC-MS/MS for highly sensitive and specific quantification
Validation of molecular biomarkers focuses on making sure the assay is reliable and reproducible. Key aspects include:
- Accuracy: Does the test measure the true value?
- Precision: Are results consistent across repeated tests?
- Sensitivity: Can it detect low-abundance biomarkers?
- Specificity: Does it avoid cross-reactivity with similar molecules?
- Stability: Does the biomarker remain stable during storage and processing?
Validating molecular biomarkers can look like running the assay on different sample batches to ensure consistent results. It could look like running the same samples in different labs to ensure consistency (also known as inter-laboratory validation). Another good practice when validating molecular biomarkers is to assess matrix effects. These could be differences in analyte detectability between serum vs plasma for example.
Molecular biomarkers are most commonly used in target engagement studies, pharmacodynamic (PD) studies, and companion diagnostics.
Imaging Biomarkers
Imaging biomarkers measure changes in biological processes that can be detected inside the body. They use medical imaging technologies like Magnetic Resonance Imaging (MRI), Positron Emission Tomography (PET), and Computerized Tomography (CT) scans. Instead of identifying and quantifying individual molecules, imaging biomarkers measure things like tumor size, changes in brain activity, or blood flow.
Validation of imaging biomarkers looks a little different than traditional biomarker validation. Since the "assays" in this case are expensive machines, it's important to validate not just the assays themselves but also ensure that image analysis is standardized. Some things to consider include:
- Reproducibility: Are results consistent across different sites and machines?
- Quantification accuracy: Are imaging measurements standardized?
- Longitudinal consistency: Can changes over time be reliably detected?
To address these challenges, validation typically includes:
- Phantom calibration studies (using standardized objects)
- Multi-center trials to assess variability across sites
- Standardized imaging protocols (e.g., acquisition parameters)
- Use of centralized image analysis platforms
Imaging biomarkers are widely used in:
- Oncology (tumor response evaluation, RECIST criteria)
- Neurology (brain atrophy, amyloid imaging)
- Cardiology (blood flow, cardiac function)
Their biggest advantage is the ability to track disease progression non-invasively and in real time, but this also makes standardization and validation especially critical.
Multi-Omics & AI-Driven Biomarkers
Multi-omics biomarkers combine data from different biological layers, including:
- Genomics (DNA mutations)
- Transcriptomics (RNA expression)
- Proteomics (protein levels)
- Metabolomics (metabolic profiles)
These techniques typically use biomarker signatures, which are a combination of multiple biomarker features. Using a signature instead of a single biomarker can improve predictive strength.
Due to the complexity of assessing multiple biomarkers at once, AI and machine learning are often used. Machine learning allows researchers to:
- Identify hidden patterns in large datasets
- Select the most relevant biomarker features
- Build predictive models for treatment response or disease progression
Validation in this space goes beyond traditional methods and includes:
- Cross-validation within datasets (training vs testing sets)
- External validation using independent cohorts
- Model performance metrics (e.g., AUC, sensitivity, specificity)
- Robustness testing across different populations and data sources
One key challenge is avoiding overfitting, where a model performs well on training data but fails in real-world scenarios. That's why independent validation datasets are critical.
These biomarkers are becoming increasingly important in:
- Early drug discovery (target identification)
- Patient stratification in complex diseases
- Predictive diagnostics for personalized therapy
While still evolving, multi-omics and AI-driven biomarkers represent one of the most promising directions in modern drug development, offering a more holistic and data-driven approach to understanding disease.
Core Principles of Biomarker Validation
1. Validation of the Analysis
The first step is to make sure that the test itself works. Does it measure what it should? Is it responsive enough? Does it stay the same throughout time?
For instance, PCR assays or protein detection platforms must always give accurate results. If the measurement isn't reliable, nothing that is constructed on it will be either.
2. Clinical Validation
Once the test works, the next question is: does the biomarker have any clinical meaning?
Clinical validation examines the association of the biomarker with actual outcomes, such as disease progression or therapy response. This necessitates research conducted at many places to ensure the findings are consistent across diverse populations.
3. Validation Driven by COU
Different biomarkers need various quantities of validation. This is the Context of Use (COU), which means how they are being used.
For instance, an exploratory biomarker in first studies might require only fundamental validation. However, a biomarker used to support drug approval must meet significantly higher requirements.
This fit-for-purpose approach helps to strike a compromise between scientific rigour and practical efficiency.
4. Statistical & Modeling Approaches
Validation relies heavily on statistics. Common approaches for assessing predictive power include ROC curves, cross-validation, and sequential testing.
Machine learning is also becoming increasingly significant since it helps researchers figure out if a biomarker really adds value to existing approaches.
Emerging Trends and Innovations
The field is evolving quickly.
AI and machine learning are making it easier to discover and validate biomarkers, especially when dealing with large, complex datasets. Multi-omics approaches are providing a more complete picture of disease biology.
Digital health tools-like wearable devices-are also opening the door to digital biomarkers, allowing continuous, real-time monitoring of patients.
At the same time, validation strategies are becoming more flexible. The fit-for-purpose approach allows researchers to adapt validation requirements based on the stage of development, making the process more efficient without compromising quality.
Conclusion
Biomarker validation bridges the gap between research and real-world application. By rigorously testing biomarkers to ensure they're reliable and clinically meaningful, we can feel confident using them to inform decisions made during clinical trials and throughout drug development.
And while validation itself is crucial, it's the enablement of biomarker discovery and development that will have the greatest impact on drug development success. Advancements in AI, multi-omics, and digital health will only increase the prevalence of biomarkers in medicine, making truly personalized treatment possible.
Creative Bioarray Relevant Recommendations
| Products & Services | Description |
|---|---|
| PK/PD Biomarker Analysis | Creative Bioarray offers comprehensive PK/PD biomarker analysis to optimize dosing, predict efficacy, ensure safety, and accelerate drug development. |
Reference
- Del Boccio P, Rossi C, et al. Integration of metabolomics and proteomics in multiple sclerosis: From biomarkers discovery to personalized medicine. Proteomics Clin Appl. 2016. 10(4):470-84..