Double Staining Apoptosis Assay (Hoechst33342/PI)
Introduction
The double staining apoptosis assay provides a rapid and convenient method for the compacted state of chromatin in apoptotic cells.
Propidium iodide (PI) is used as a DNA stain by intercalating between the bases with little or no sequence preference. Once the dye binds to nucleic acid, its fluorescence is enhanced 20 to 30 fold. PI is excited at 488 nm and its maximum emission wavelength is 617 nm. As a membrane impermeable dye that generally excluded from viable cells, it is commonly used as a counterstain in multicolor fluorescent techniques to distinguish between late apoptotic cells and normal cells in a population.
Hoechst stains are cell-permeable nuclear counterstain that emit blue fluorescence when combined with dsDNA. These bisbenzimidazole derivatives bind into the minor groove of DNA with adenine thymine (AT) selectivity. Hoechst dyes are very sensitive to DNA conformation and chromatin status in cells and are therefore used to detect the gradations of nuclear damage, such as distinguishing condensed pycnotic nuclei in apoptotic cells.
The staining pattern resulted from the simultaneous use of these dyes makes it possible to distinguish normal, apoptotic, and dead cell populations by flow cytometer and fluorescence microscopy.
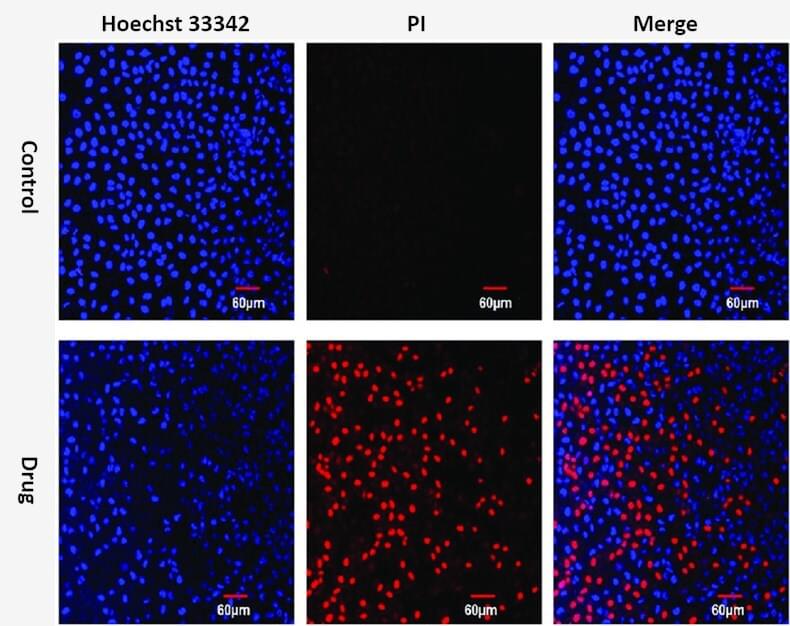 Figure 1. Hoechst 33342 and PI double staining in MCF-7 cells.
Figure 1. Hoechst 33342 and PI double staining in MCF-7 cells.
Key Features
- Easy to perform: simple and rapid procedure to perform.
- Fast and quick: all of the procedure is in 20 minutes.
- Versatile: directly analyze normal, apoptotic, and dead cells by flow cytometry and fluorescence microscopy.
Protocol
- Induce apoptosis by a desired method. A negative control should be prepared without inducing reagent.
- Harvest the cells, wash with cold PBS or culture medium and adjust the cell density to 1 × 106 cells/mL.
Note: Directly collect suspension cells by centrifugation. Otherwise, inhered cells should be digested firstly to use. - Add 10 μL of Hoechst dye to each of the cell suspension and mix thoroughly. Incubate the cells at 37°C for 5-15 minutes.
- Centrifuge the cells at 1,000 rpm for 5 minutes at 4°C and discard the supernatant.
- Resuspend cells in 1000 µL of 1X PBS.
- Add 5 μL of PI to each of cell suspension and mix thoroughly. Incubate the cells at room temperature for 5-15 minutes.
- After the incubation, analyze the stained cells by flow cytometry immediately, using UV/488 nm dual excitation and measuring the fluorescence emission at 460 nm emission of Hoechst33342 and 617 nm emission of PI.
Note: 1. The Hoechst 33342/PI are suspected carcinogens, operation with gloves, protective clothing, and eyewear.
2. Hoechst 33342/PI is sensitive to light, so all experiment process need keep away from light.
References
- Cao H. et al.; Synthesis, cytotoxicity and antitumor mechanism investigations of polyoxometalate doped silica nanospheres on breast cancer MCF-7 cells. PLOS ONE, 2017, 12 (7): 1-18.
- Prasad R. et al.; In vitro anticancer activities of ethanolic extracts of dendrobium crepidatum and dendrobium chrysanthum against T-cell lymphoma. J Cytol Histol, 2016, 7:432.
Cell Services:
Cell Line Testing and Assays:
Explore Other Options