Undifferentiated iPSC Detection
A key concern in iPSC-based therapeutic applications is the safety of the cell products to be transplanted into patients. Although the in vivo teratoma formation assay is an effective method to directly assess the tumorigenicity of undifferentiated cells, it is also costly and time-consuming. Now, Creative Bioarray presents a quick and cost-effective assay to detect residual undifferentiated iPSCs amongst direct differentiated cells and cells intended to be used in transplantation activities.
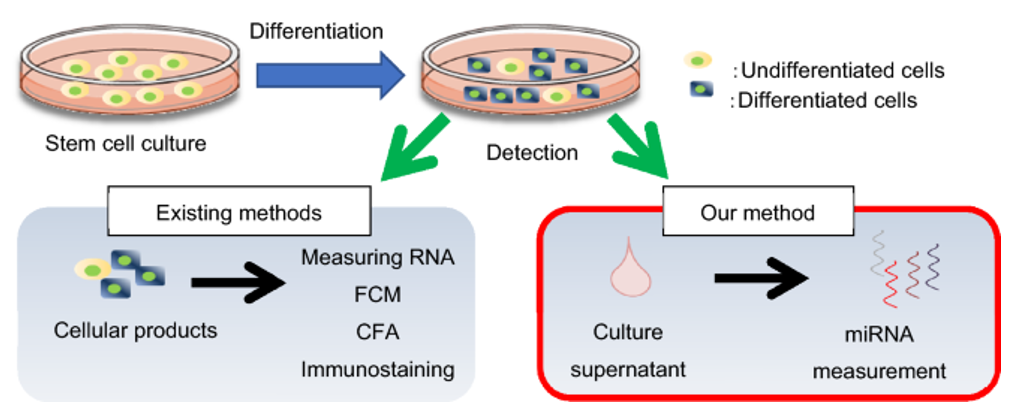
We detect the undifferentiated iPS cells by measuring miRNA markers in the cell culture supernatants, which is non-destructive, but more sensitive than other testing methods. This technology could also be used for early cell selection, contributing to the improvement of production efficiency.
Service features:
- More sensitive than the conventional Lin28A method
- Non-destructive
- Measurement of differentiation efficiency
- Evaluation of tumor formation risk
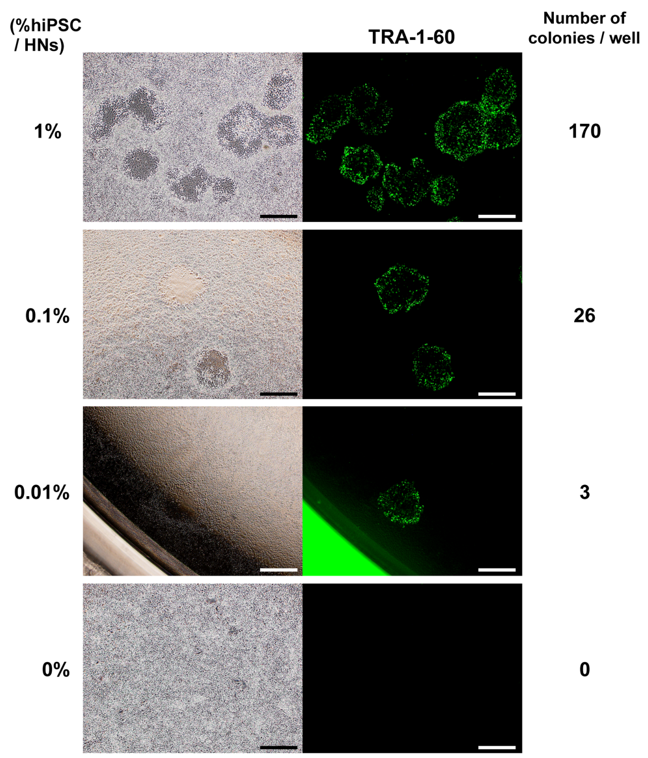 Figure 1. Detection of hiPSCs spiked into human neurons.
Figure 1. Detection of hiPSCs spiked into human neurons.
Quotation and ordering
We have extensive experience in developing safety analysis of regenerative medicine products. To discuss any of these models further or to discuss the possibility of developing alternative models, please do not hesitate to contact us.
Explore Other Options
For research use only. Not for any other purpose.
Services
-
Cell Services
- Cell Line Authentication
- Cell Surface Marker Validation Service
-
Cell Line Testing and Assays
- Drug-Resistant Cell Models
- Cell Viability Assays
- Cell Proliferation Assays
- Cell Migration Assays
- Soft Agar Colony Formation Assay Service
- SRB Assay
- Cell Apoptosis Assays
- Cell Cycle Assays
- Cell Angiogenesis Assays
- DNA/RNA Extraction
- Cellular Phosphorylation Assays
- Stability Testing
- Sterility Testing
- Endotoxin Detection and Removal
- Phagocytosis Assays
- Ferroptosis Assay
- Cell-Based Screening and Profiling Services
- 3D-Based Services
- Custom Cell Services
- Cell-based LNP Evaluation
-
Stem Cell Research
- iPSC Generation
- iPSC Characterization
-
iPSC Differentiation
- Neural Stem Cells Differentiation Service from iPSC
- Astrocyte Differentiation Service from iPSC
- Retinal Pigment Epithelium (RPE) Differentiation Service from iPSC
- Cardiomyocyte Differentiation Service from iPSC
- T Cell, NK Cell Differentiation Service from iPSC
- Hepatocyte Differentiation Service from iPSC
- Beta Cell Differentiation Service from iPSC
- Brain Organoid Differentiation Service from iPSC
- Cardiac Organoid Differentiation Service from iPSC
- Kidney Organoid Differentiation Service from iPSC
- GABAnergic Neuron Differentiation Service from iPSC
- Undifferentiated iPSC Detection
- iPSC Gene Editing
- iPSC Expanding Service
- MSC Services
- Stem Cell Assay Development and Screening
- Cell Immortalization
-
Molecular Biology Solutions
- Chromosome & Genomic Analysis
-
Cytogenetics & Molecular Cytogenetics Analysis
- Fluorescent In Situ Hybridization (FISH)
- In Situ Hybridization (ISH) & RNAscope
- ImmunoFISH (FISH+IHC)
- I-FISH
- Splice Variant Analysis (FISH)
- RNA FISH in Plant
- mtRNA Analysis (FISH)
- Digital ISH Image Quantification and Statistical Analysis
- Telomere Length Analysis (Q-FISH)
- Telomere Length Analysis (qPCR assay)
- Droplet Digital PCR (ddPCR)
- QuantiGene Plex Assay
- Probe Development & Quality Control
- ISH/FISH Analysis for Therapeutic R&D
- Cell Line Characterization
- Pathogen & Microbial Analysis
- Histology Services
- Exosome Research Services
- Drug Metabolism and Pharmacokinetics (DMPK)
-
Safety Evaluation Services
- High-Content Cytotoxicity Screening
- High-Throughput Toxicity Screening
- In Vitro Cardiotoxicity
- In Vitro Genotoxicity
- Hepatotoxicity
- In Vitro Neurotoxicity
- In Vitro Nephrotoxicity
- In Vitro Dermal Toxicology
- Ocular Toxicity
- In Vitro Cytotoxicity
- Endocrine Disruption Screening Assay
- In Vivo Toxicity Study