Immortalized Cell Culture Guide
It has been well-documented that primary cells can only undergo a predetermined and finite number of cell divisions in culture, which is the biggest weakness of them. With the population doublings, primary cells enter a state where they can no longer divide called replicative senescence-cell morphology, gene expression, and metabolism all have changed. So it's important to establish immortalized cells in vitro to study cell growth, differentiation, and senescence. This concise guide is designed to help the newcomers understand the process of establishing immortalized cell lines from primary cultures.
Overview of Immortalized Cell
Immortalized cells are a population of cells from a multicellular organism due to mutation, which can escape normal cellular senescence and keep undergoing division. Thus, this kind of cells can grow in vitro for prolonged periods. The mutations required for immortality can occur naturally or be intentionally induced for experimental purposes. Immortal cell lines are a powerful tool for the biological investigator to research the biochemistry and cell biology of multicellular organisms, which plays an important role in the study of the biology of cell growth, differentiation, and senescence. And they have been applied in the research of cell biology, immunology, hematology, cancer biology, toxicology, and molecular biology.
Why Do We Need Immortalized Cells?
Firstly, primary cells will reach senescence after limited generation. The process of frequently re-establish fresh cultures from explanted tissue is tedious and it can also add significant variation from one preparation to another. Using immortalized cell can guarantee the experiment materials' consistent.
Next, in addition to the capacity of extended proliferation, the immortalized cells possess similar or identical genotype and phenotype to their parental tissue. Additionally, in many studies hTERT immortalized cells have been induced to become differentiated cell types, exhibiting tissue-specific features, differentiation-specific proteins, and forming structures that resemble those formed in vivo. All the mentioned above make immortalized cells become vital factor in cell biology research.
Strategies for Creating Immortal Cells
Here we will discuss various ways to overcome the hayflick limit and induce immortality in cultured cells.
- Spontaneously Mutation
Some primary cell may be mutated and break the limited of lifespan in the process of dividing and multiplying. Such cells will be collected for expanded cultivation and become the immortalization cells, which transformation is called spontaneously mutation. But this method is inefficient and the cells will transform into the tumor cells in most cases. So, tumor cells would be the best example of spontaneously immortalized cells, which may have undergone genetic changes to resist senescence and are immortal.
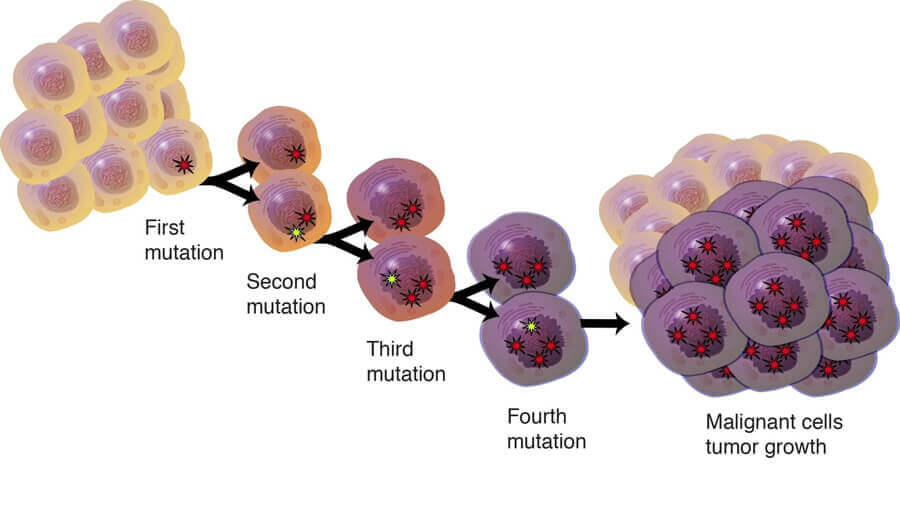
But it's worth noting that there is a basic difference between tumor cells and immortalized cells: tumor cells have many classical characteristics such as losing contact inhibition, low adhesive ability, and apoptosis inhibition while immortalized cells remain their normal genotype and phenotype.
- Introduce a Viral Gene that Overrides the Cell Cycle
Many viral genes can affect the cell cycle, so they are able to establish immortalization by removing the biological brakes on proliferative control. One method is to use the simian virus 40 (SV40) T-antigen, to induce immortalization. SV40 T-antigen has been shown to be the simplest and most reliable agent for the immortalization of many different cell types and the mechanism of SV40 T-antigen in cell immortalization is relatively well understood. One example is HEK293T (also known as 293T).
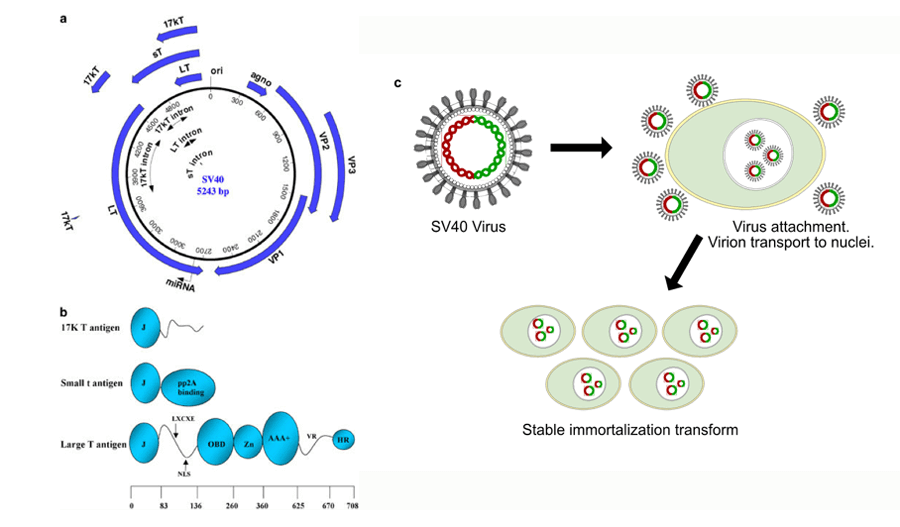 Figure 1. (a) Structure of SV40 genomic DNA is composed of three elements: the early and late coding units and the regulatory region. (b) Several domains and motifs make up the SV40 T antigens. (c) A brief overview of the process of SV40 induced immortalized transformation. (Ahuja D,2005)
Figure 1. (a) Structure of SV40 genomic DNA is composed of three elements: the early and late coding units and the regulatory region. (b) Several domains and motifs make up the SV40 T antigens. (c) A brief overview of the process of SV40 induced immortalized transformation. (Ahuja D,2005)
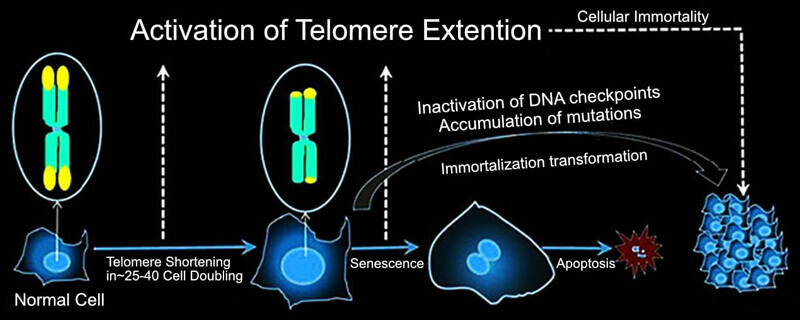
- Expression of Telomerase Reverse Transcriptase (TERT) Protein
Telomerase is a ribonucleoprotein that is able to extend the DNA sequence of telomeres, which make the cells to undergo infinite cell divisions through abating the senescence process. This protein is inactive in most somatic cells, but when TERT is exogenously expressed, the cells are able to maintain sufficient telomere lengths to avoid replicative senescence. Human telomerase reverse transcriptase (hTERT) is the most popular approach to cell immortalization at present.
- Combining Suppressor Cell Cycle and Telomerase Expression
In some cell types, using only one immortalization method may not be able to yield immortal cells. Therefore, depending on different cell lines it would be better to combine suppressor cell cycle and expression of hTERT to immortalize a larger number of cells.
In all methods mentioned above, expression of hTERT is recommended.
Compared with traditional amortization cells especially at high passage numbers and when oncogenes are used, hTERT immortalized cells faithfully represent the physiological properties of normal cells in vivo. The following is a summary of the commonly observed properties of cells immortalized by the ectopic expression of hTERT.
- Nonmalignant
- Normal cell cycle controls, functional p53 and pRB checkpoints
- Contact inhibited
- Anchorage dependent
- Retain normal growth responses to serum and mitogens
- Require growth factors for proliferation
- Possess a normal karyotype
| Primary Cells | hTERT-Immortalized | Onco, Viral-Immortalized | Continues | |
| Mimic in vivo Tissue Phenotype | √ √ √ √ | √ √ √ | √ √ | √ |
| Karyotypic Stability | Diploid | Diploid/Pseudodiploid | Pseudodiploid/Aneuploid | Aneuploid |
| Proliferative Capacity | √ | √ √ √ | √ √ √ | √ √ √ |
| Supply | √ | √ √ √ | √ √ √ | √ √ √ |
| Inter-Experimental Reproducibility | Low | Good | Good | Good |
| Cost | HIGH | MEDIUM | LOW | LOW |
| Ease of Use | √ | √ √ | √ √ | √ √ √ |
Table 1. Comparison between hTERT Immortalized Cells, Primary Cells, Oncogene/Viral Immortalized Cells and Continuous Cell Lines.
Getting Started with Creative Bioarray
With more than 10 years of experience in cell immortalization, experts at Creative Bioarray have developed multiple cell immortalization products. One of our featured product is hTERT induced immortal service, in addition to plasmids, retroviral, lentiviral and adenoviral vectors, p53 and RB, siRNAs, and SV40 T-antigens are also available. Meanwhile, we also provide ready-to-use recombinant retroviruses, lentivirus, and adenoviruses which have been incorporated with these genes in order to make your project simpler and easier.
Except for providing cell immortalization services, we also offer commercial immortalization cells for global clients. Our immortalization cell bank owns more than 160 different cell types from human and over 13 types of animals.
Technical Support
In the technical bulletin, we provide the most current methods for the immortalization of cells, as well as specific guidelines for the immortalization of kidney, and thyroid epithelia, hepatocytes, fibroblasts, chondrocytes, lymphocytes, astrocytes, and human umbilical vein endothelium. Each method is accompanied by:
- Step-by-step protocols
- Detailed instructions for reagent preparation
You can download these technical resources on our website or contact our online customer service to obtain them.
Creative Bioarray dedicates to providing the most up-to-date in vitro methods and safety procedures for the immortalization of various cell types. We are capable of accommodate your needs and achieving desired goals of the study. If you want to know more about our services and products, please feel free to contact us. We look forward to working with you in the future.
References
- Stewart S A .; et al. Telomerase contributes to tumorigenesis by a telomere length-independent mechanism. Proceedings of the National Academy of Sciences. 2002, 99(20): 12606-12611.
- Kirchhoff C.; et al. Immortalization by large T-antigen of the adult epididymal duct epithelium. Molecular and cellular endocrinology. 2004, 216(1): 83-94.
- Ahuja D.; et al. SV40 large T antigen targets multiple cellular pathways to elicit cellular transformation. Oncogene. 2005, 24(52): 7729-7745.
- Macieira-Coelho A. Cell Immortalization. Springer Berlin Heidelberg. 2000.