Drug Efficacy Evaluation

The progress from drug discovery to its commercialization is complex, lengthy and expensive. Before a new drug comes to market, it should be tested and evaluated from several ways as follows:
Drug efficacy evaluation provides an analytic framework for making better decisions about a new drug’s appropriate place in therapy.
Many experimental cancer models have been developed to investigate carcinogenesis, cancer progression, metastasis, and other aspects in oncology and these models turned out to be useful in the efficacy evaluation and the safety prediction of oncology drugs.
Creative Bioarray offers several assays to evaluate a compound's effects on tumor cells in vitro, including cell cycle assays, proliferation, apoptosis, migration & invasion, cytotoxicity, and cell-based drug discovery services. We also perform a number of evaluation assays in vivo. Using various tumor cell lines, primary cultured cells, and animal tumor models, we examine causes of cell viability and metabolism by compounds or drugs to investigate mechanisms of efficiency and toxicity of pharmaceuticals. We conduct experiments using multiple advanced analysis systems and technologies, such as flow cytometers, laser scanning cytometers, laser capture microdissection systems, real-time PCR systems, and DNA microarray analysis systems.
Within decades of research experience on drug efficacy evaluation, Creative Bioarray is always ready to provide study design and experiment for customers to facilitate their research on drug discovery and screening.
Advantages
- Get robust results with a fast turnaround time
- Obtain rich data generated by our dedicated team of scientists
- Satisfy different demands using various cell lines and diversified advanced analysis systems
Workflow

Study examples
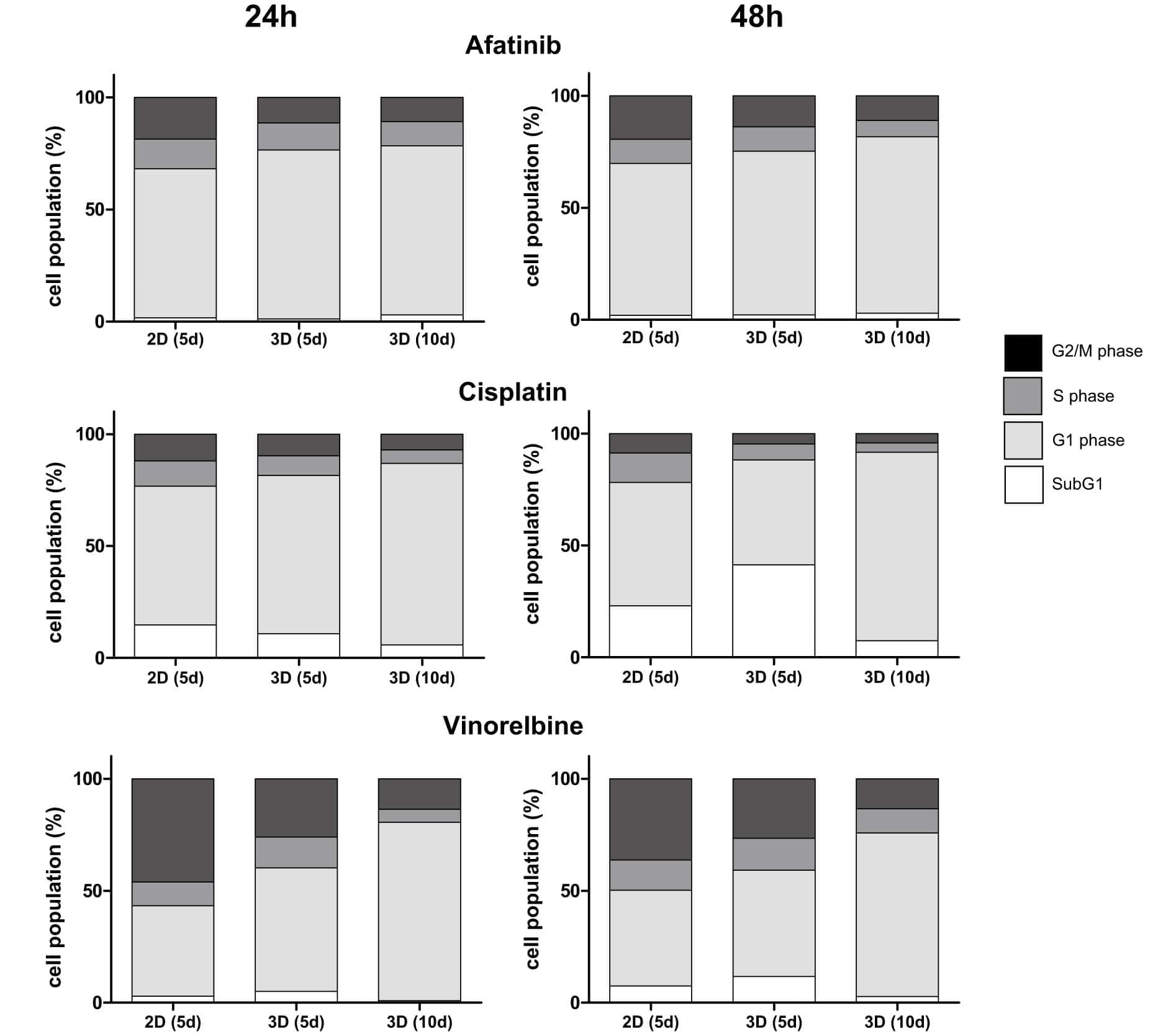
Fig.1 Determination of cytostatic effect: cells and microtissues are treated with increasing doses of afatinib, cisplatin and vinorelbine for 24 and 48 h.
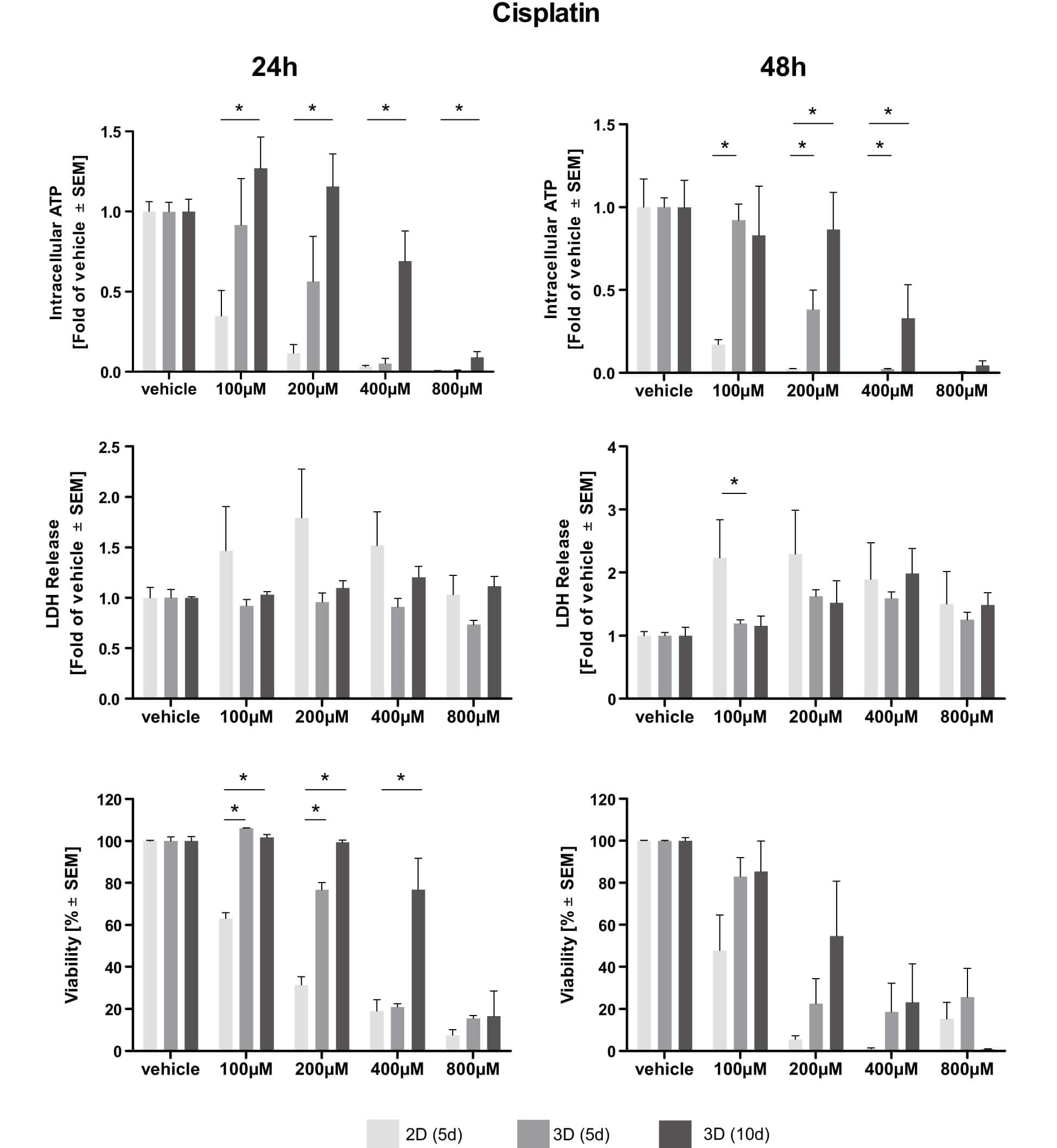
Fig.2 Dose–response curves of cisplatin
Quotation and ordering
If you have any special needs in Drug Efficacy Evaluation, please contact us for this special service. Let us know what you need and we will accommodate you. We look forward to working with you in the future.
References
| 1. | Peglere, S.; Underhill, J. Evaluating the safety and effectiveness of new drugs. American Family Physician. 2010, 82(1): 53-57. |
| 2. | Huber, J.M.; et al. Evaluation of assays for drug efficacy in a three-dimensional model of the lung. J Cancer Res Clin Oncol. 2016, 142: 1955–1966. |
| 3. | Hayakawa, Y.; et al. Report on the use of non-clinical studies in the regulatory evaluation of oncology drugs. Cancer Sci. 2016, 107: 189–202 |
Explore Other Options