Why Oncology Organoids Fail? How to Build Models That Work
Let's be honest: the old ways of testing cancer drugs are failing us. For decades, we've relied on cells grown flat on plastic dishes (2D) or human tumors shrunk into mice (PDX). The first is too simple; the second is too slow, too expensive, and-let's face it-mice aren't humans.
This is where Patient-Derived Organoids (PDOs) come in. Think of them as "3D mini-tumors" grown in a lab that actually act like the patient they came from. If you're a drug developer, these are your best shot at knowing if a molecule will work before you spend millions on a clinical trial.
But "using" them isn't just about buying a kit. It's about solving real-world technical headaches. Here's the breakdown of how to make it work.
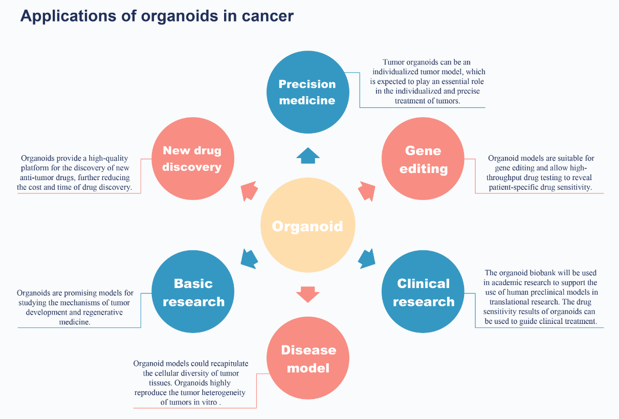
The Foundation: Solving the "Establishment" Puzzle
The biggest barrier to entry for many biotech firms is the inconsistency of organoid establishment. You cannot run a drug screen if your "seed" doesn't grow.
The Problem: Sample Quality and Heterogeneity
Most clinical samples-whether from a needle biopsy or surgical resection-are messy. They arrive contaminated with blood, necrotic (dead) tissue, and "normal" stromal cells that can easily outgrow the tumor cells. If you follow a generic protocol, your success rate might stay stuck at 30-40%.
The Solution: Standardized "Cold-Chain" and Gentle Dissociation
To push success rates toward 80% or 90%, the process must be treated like a surgical procedure:
- Ischemia Management: The clock starts the second the tissue is removed. We use specialized preservation media and validated transport containers to keep the cells in a state of "metabolic suspended animation" at 4°C.
- The "Hybrid" Method: Avoid aggressive, long-term enzymatic digestion (like using high-dose Trypsin for an hour). We've found that a combination of mechanical "mincing" followed by a short, gentle enzymatic cocktail preserves the delicate surface receptors and stemness of the tumor cells.
- Cleaning the Slate: Always include a dedicated step to lyse red blood cells and inhibit fibroblasts early on. A "clean" culture in the first 72 hours is the difference between a usable model and a wasted biopsy.
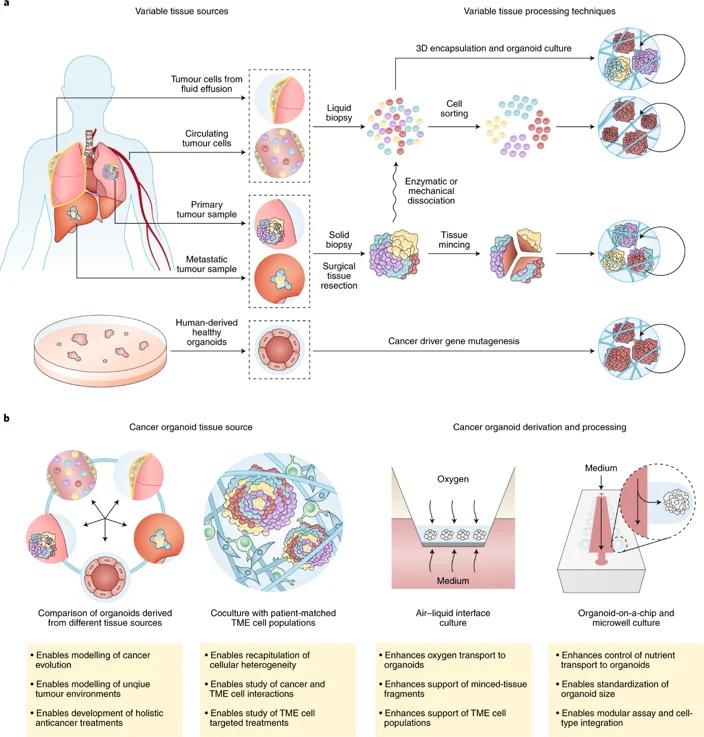
Drug Screening: From "Pretty Pictures" to Reliable Data
Once you have the models, you want to test your compounds. But 3D drug sensitivity testing (DST) is significantly more complex than 2D testing.
The Problem: Penetration and Signal Noise
In a 2D dish, every cell is exposed to the drug instantly. In an organoid, the drug must penetrate a thick basement membrane (Matrigel) and multiple layers of cells. Furthermore, standard metabolic assays often provide a "smeared" average that can hide resistant sub-clones.
The Solution: High-Content Imaging (HCI) and Kinetic Monitoring
- Standardizing the "Ball": You cannot compare a 50μm organoid to a 500μm one. We utilize automated "cell strainers" and liquid handling robotics to ensure every well starts with a uniform distribution of organoid "seeds."
- Seeing is Believing: Instead of just measuring a light signal at the end of the week, use High-Content Imaging. This allows you to track the morphology. Is the drug killing the cells, or is it just stopping them from growing (cytotoxic vs. cytostatic)? Is the core of the organoid dying while the shell stays alive?
- The AUC Metric: Stop obsessing over IC50 alone. In heterogeneous oncology, the Area Under the Curve (AUC) provides a much more robust picture of how a drug performs across a range of doses, especially when dealing with drugs that have a narrow therapeutic window.
The "Neighborhood": Adding the Immune System
A "naked" organoid-consisting only of cancer cells-is like a soldier without an army. To test modern immunotherapies, you need the neighborhood.
The Problem: Testing Immunotherapies (IO)
Standard PDOs don't contain T-cells. Therefore, you cannot test PD-1/PD-L1 inhibitors or CAR-T therapies using traditional 3D cultures.
The Solution: Reconstituted Autologous Co-Cultures
This is where the real "CRO magic" happens. We don't just culture the tumor; we co-culture it with the patient's own immune cells (PBMCs).
- The Workflow: We isolate the tumor-infiltrating lymphocytes (TILs) or PBMCs from the same patient. We "prime" these immune cells and introduce them to the organoids in a controlled ratio.
- Functional Readouts: We track T-cell infiltration. A successful drug candidate should not only kill the tumor but also increase the speed and depth at which T-cells penetrate the 3D organoid structure.
- Adding the "Shield": To simulate real-world resistance, we can also add Cancer-Associated Fibroblasts (CAFs). These cells create the physical "stiffness" that prevents drugs from reaching the tumor. Testing your drug in a "CAF-rich" model provides a brutal, realistic look at how it will perform in a real patient.
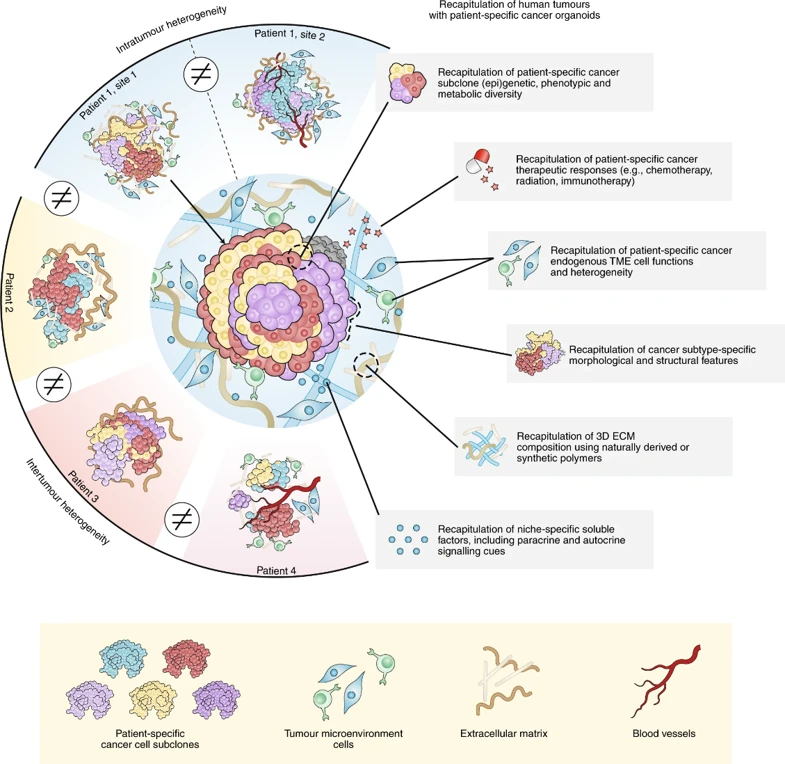
Strategic Integration: Where Does it Fit in the IND Pipeline?
You don't need organoids for everything, but you need them for the right things.
Lead Optimization (The Filter)
If you have 50 potential molecules, running them through 50 PDX mice is a financial nightmare. Running them through 50 patient organoid models is a smart investment. It allows you to rank-order your candidates based on real human tissue response before you ever touch a living animal.
Indication Expansion (The Compass)
If your drug works in Lung Cancer, will it work in Ovarian Cancer? Instead of launching a blind clinical trial, you can run a "basket screen" across hundreds of diverse organoid models to identify which specific mutations or cancer types are most sensitive to your drug.
Regulatory Readiness: Speaking the Language of the FDA
The FDA Modernization Act 2.0 has opened the door for "non-animal" data to support IND (Investigational New Drug) applications. But the data must be "clean."
The CRO Responsibility: Validation and Compliance
To use organoid data for a regulatory filing, you need more than a graph. You need:
- Genomic Stability Reports: Evidence that the organoid hasn't mutated into something unrecognizable after 10 passages.
- Batch Consistency: Showing that your Matrigel and growth factors are consistent so that the results today match the results next month.
- Standardized QC: Morphological analysis, STR profiling, and IHC staining to prove the model is what you say it is.
Conclusion: Getting it Right the First Time
At the end of the day, organoids are the most sophisticated "biological simulator" we have. They allow us to fail fast, fail cheap, and move the winners forward with confidence.
As a CRO, our goal isn't just to grow cells in a dish; it's to provide a predictive platform. By solving the technical hurdles of sample handling, automation, and microenvironment complexity, we turn organoids from a "cool science project" into a cornerstone of modern oncology drug discovery.
The future of oncology isn't just about finding any drug; it's about finding the right drug for the right patient. And the "how-to" starts here, in the 3D architecture of the organoid.
Creative Bioarray Relevant Recommendations
| Products & Services | Description |
|---|---|
| PDO-based Drug Screening Services | Creative Bioarray's PDO-based drug screening services using patient-derived organoids to evaluate drug efficacy, resistance, and personalized treatment with high clinical relevance. |
References
- Fang Z, Li P, et al. The role of organoids in cancer research. Exp Hematol Oncol. 2023. 12, 69.
- LeSavage B L, Suhar R A, et al. Next-generation cancer organoids. Nat. Mater. 2022. 21, 143-159.