Troubleshooting Common Issues in Drug Toxicity Testing
Drug toxicity testing is an important consideration during preclinical drug development. Evaluating the toxicity of your next drug candidate is one of the most important factors that will determine if your compound is safe to test in patients. During toxicity testing, researchers perform numerous in vitro and in vivo experiments to assess compound toxicity, identify its maximum tolerated dose, and determine its mechanism of action.
There are however several issues that can interfere with toxicity studies. Everything from experimental variation and model limitations to poor pharmacokinetics (PK) can impact your toxicity studies. Failure to recognize and address these problems can lead to false toxicity results or missed safety signals.
Therefore, it is important that researchers know common drug toxicity testing issues and how to troubleshoot them. Below we highlight some common problems that occur during toxicity studies and provide practical tips for troubleshooting.
Common Challenges in Drug Toxicity Testing
Drug toxicity studies involve complex biological systems, and multiple factors can influence the reliability of experimental results. The following table summarizes several common challenges and their potential impact on toxicity evaluation.
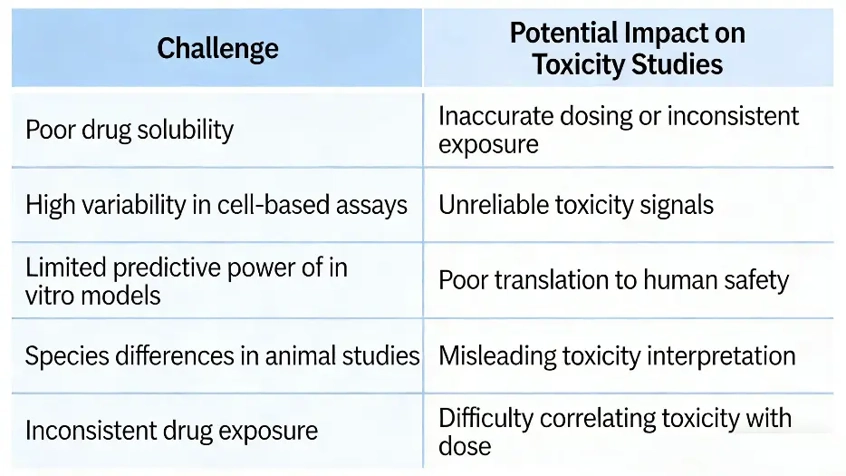
Identifying and addressing these issues early can significantly improve the accuracy and reproducibility of toxicity testing results.
Problem 1: Poor Drug Solubility and Formulation Issues
Poor compound solubility is one of the most common technical problems encountered during toxicity studies. Many small molecule drug candidates have poor aqueous solubility. As a result, this creates several dosing challenges and limits exposure in vitro and in vivo.
Poor solubility can impact cell-based assays by decreasing effective drug concentration due to precipitation within culture media. In animal models, poor formulation can limit absorption and systemic exposure.
To overcome poor solubility issues you can:
- Optimizing drug formulation using suitable solvents or excipients
- Adjusting pH conditions to improve compound solubility
- Applying advanced delivery approaches such as lipid-based formulations or nanoparticles
- Verifying actual drug exposure through pharmacokinetic measurements
Careful formulation development and exposure monitoring are essential for ensuring that toxicity results accurately reflect the compound's biological activity.
Problem 2: High Variability in In vitro Toxicity Assays
Cell-based toxicity assays are a popular choice during early drug discovery as they allow for high-throughput safety screens. However, these assays can sometimes produce highly variable results, which complicates data interpretation.
Several factors may contribute to assay variability:
- Differences in cell passage number or cell line stability
- Variations in culture conditions such as media composition or incubation time
- Batch-to-batch differences in reagents or assay kits
- Manual handling errors during experimental procedures
To reduce variability and improve assay reproducibility, researchers can implement several best practices:
- Standardizing cell culture and assay protocols
- Using well-characterized and validated cell lines
- Including appropriate positive and negative controls
- Increasing the number of biological replicates
- Implementing automated liquid handling or high-throughput screening platforms
Establishing robust experimental procedures helps ensure that toxicity signals observed in vitro are consistent and biologically meaningful.
Problem 3: Limited Predictive Power of Traditional Cell Models
In vitro cell models have been the standard during toxicity testing for decades. However, because these cells are often grown on flat plastic surfaces, they can lose crucial physiological characteristics. As a result, these conventional models often lack the predictive power needed to anticipate toxicity in humans.
Several limitations of conventional cell culture systems include:
- Lack of three-dimensional tissue architecture
- Limited cell-cell and cell-matrix interactions
- Reduced metabolic activity compared with human organs
Due to these limitations, toxicity data generated from simple in vitro models often fails to translate into the clinic.
Fortunately, there are newer in vitro models that better mimic human physiology. These models include:
- Three-dimensional (3D) cell culture
- Organoids
- Microphysiological systems such as organ-on-a-chip
With recent advances in technology, researchers can now generate more predictive toxicology data in vitro.
Problem 4: Species Differences in Animal Toxicology Studies
Animal toxicology studies are vital for understanding the effects a compound has on a living organism. A key consideration in drug toxicity research is that animal models and humans aren't physiologically identical, potentially skewing study results.
Differences in metabolism, expression of drug targets, immune function, and biological pathways can create disparity in the way a drug behaves in animals versus humans. A drug might appear safe in animal studies, only to prove toxic in human clinical trials.
To help overcome species differences, researchers can:
- Choose an animal model that more closely mimics human metabolism
- Perform studies in both rodent and non-rodent species (if required by regulatory agencies)
- Combine animal data with human relevant in vitro models
- Perform translational toxicology to compare animal and human exposure
- By integrating multiple experimental approaches you can increase your confidence that a compound will be safe in humans.
Problem 5: Off-Target Toxicity and Unclear Mechanisms
Sometimes you may identify undesirable toxicity that is caused by off-target interactions. If this occurs, there can often be confusion around the mechanism that is responsible for the toxicity.
Mechanistic toxicology studies are helpful to better understand the biomolecular process responsible for a toxic response. There are several techniques available to help you identify the mechanism of toxicity. Some of these techniques include:
- Gene expression profiling
- Proteomic or metabolomic analysis
- Pathway-based biomarker studies
- High-content cellular imaging
By identifying the molecular processes responsible for toxicity, scientists can determine whether the effect is dose-dependent, reversible, or target-related, which can guide further drug optimization efforts.
Problem 6: Inconsistent Drug Exposure in Toxicology Studies
Another frequent challenge during toxicity studies is inconsistent drug exposure. During drug development, it is critical to maintain consistent drug exposure across all your studies. Without consistent exposure, you will not be able to correlate toxicity with dose.
Several factors can influence drug exposure levels:
- Poor formulation or instability of the compound
- Rapid metabolism or clearance in vivo
- Inaccurate dosing procedures
- Differences in absorption or distribution
To address these issues, researchers often perform pharmacokinetic (PK) and toxicokinetic (TK) analyses alongside toxicology studies. These analyses measure drug concentrations in biological samples and help determine how exposure relates to observed toxicity.
Key parameters typically evaluated include:
- Maximum plasma concentration (Cmax)
- Area under the concentration-time curve (AUC)
- Drug accumulation during repeated dosing
Integrating exposure data with toxicology findings enables a clearer understanding of dose-response relationships and safety margins.
Best Practices for Improving Toxicity Testing Reliability
While there are several challenges with toxicity testing, you can overcome many of these issues by following some simple best practices.
Key recommendations include:
- Designing well-controlled and reproducible experiments
- Standardizing protocols across laboratories and studies
- Using multiple complementary biological models
- Integrating pharmacokinetic and toxicokinetic data
- Conducting early risk assessments during lead optimization
Applying these strategies helps researchers generate robust and reproducible safety data, which is essential for successful drug development.
Troubleshooting Drug Toxicity Tests: What's Next?
Drug toxicity testing is important for the drug discovery process but can be difficult. We discussed some common problems that occur during toxicity studies and how you can troubleshoot them.
Remember, some of the best practices include designing rigorous and well-controlled experiments, using multiple models, and integrating PK/TD data.
If you need support with your preclinical toxicity studies, we provide characterization assays to determine drug-induced toxicity. Contact us today to learn how we can help optimize your drug development pipeline.
Creative Bioarray Relevant Recommendations
| Products & Services | Description |
|---|---|
| Safety Evaluation Services | With years of expertise, Creative Bioarray serves as a trusted partner in safety evaluation, specializing in preclinical toxicology testing. Our comprehensive testing platform supports applications throughout all stages of product development. |
| In Vivo Toxicity Study | Creative Bioarray's GLP toxicology studies include administration routes ranging from acute to chronic. With internal resources, we can also provide a full range of toxicological assessments. |