In Situ Detection of CAR-T Cells & Oncolytic Viruses
CAR-T cells have yielded unprecedented efficacy in B cell malignancies, most remarkably anti-CD19 CAR-T cells for B cell acute lymphoblastic leukemia (B-ALL) with up to a 90% complete remission rate.
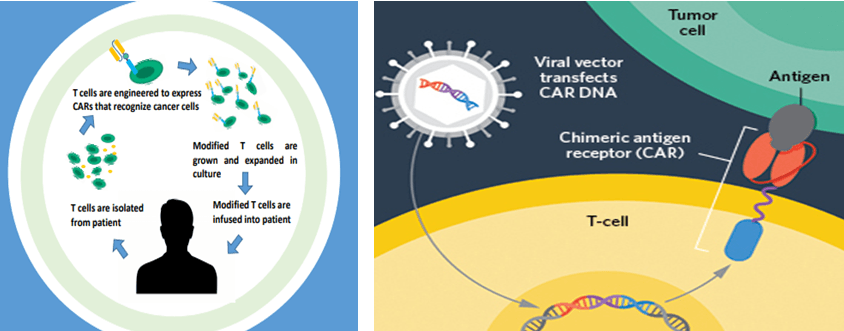
The ability to rewire our own immune system to fight cancer has certainly created huge expectations. After the success, more and more preclinical experiments and Phase I-III trials will be performed on large groups. The distribution of CAR-T cells and Oncolytic Viruses in the tumor and in the body is important for assessing the antitumor effect and safety of the injected CAR-T or Oncolytic Viruses in vivo, while observing the antitumor effect. Because of the heterogeneity of the tumor, the in situ localization and distribution of CAR-T cells and oncolytic virus in tumor cells and tumor microenvironment can reflect the mechanism and application prospect of the treatment method.
As CAR-T and oncolytic viruses themselves carry their own unique nucleic acid sequences, RNA specific probes designed for these regions can effectively detect the distribution of these cells or viruses in situ. So RNA ISH is a unique and effective tool for detecting CAR T cells and viruses in situ.
With years of professional experience in the RNA ISH Probe and ISH Service field, Creative Bioarray's streamlined process can offer timely and dependable in situ detection of CAR-T and Oncolytic Viruses results, giving you the flexibility to focus on other research priorities.
Applications
- In Situ detect the distribution of CAR-T cells/Oncolytic Viruses in tumor cells or tissues
- Assess the antitumor effect and safety of the injected CAR-T or Oncolytic Viruses in vivo
- Confirm CAR expression in FFPE tissue
- Quantify the CAR expression in FFPE tissue
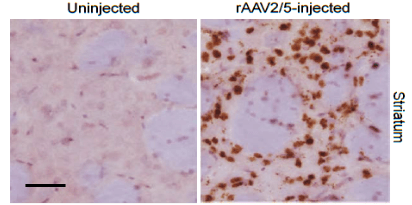 Figure 1. The in situ localization and distribution of rAAV Detection by RNA ISH. Scale bars= 5μm
Figure 1. The in situ localization and distribution of rAAV Detection by RNA ISH. Scale bars= 5μm
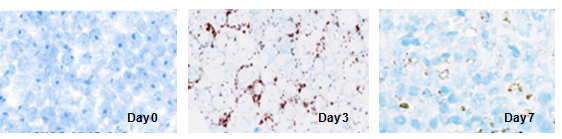 Figure 2. The in situ localization and distribution of CAR T Cells detection by RNA ISH.
Figure 2. The in situ localization and distribution of CAR T Cells detection by RNA ISH.
Features
- Accurate-In Situ Detection Service-Custom design your probe
- Value – We focus on the quality of our service and all supported by competitive pricing
- Efficiency – We are able to provide the fastest turnaround time of any supplier in the industry
Quotations and ordering
Our customer service representatives are available 24hr a day! We thank you for choosing Creative Bioarray at your preferred In situ Detection of CAR-T Cells & Oncolytic Viruses Services.
References
- Grabinski T M,; et al. A method for combining RNA in situ hybridization with immunohistochemistry in thick free-floating brain sections and primary neuronal cultures[J]. PLoS One, 2015, 10(3): e0120120.
- Posey A D,; et al. Engineered CAR T cells targeting the cancer-associated Tn-glycoform of the membrane mucin MUC1 control adenocarcinoma[J]. Immunity, 2016, 44(6): 1444-1454.
- Haile S T,; et al. Attenuated oncolytic virus HSV1716 increases in vivo expansion of GD2-targeting CAR T cells in murine solid tumor models[J]. AACR 107th Annual Meeting 2016; April 16-20, 2016.
Explore Other Options
For research use only. Not for any other purpose.
Services
-
Cell Services
- Cell Line Authentication
- Cell Surface Marker Validation Service
-
Cell Line Testing and Assays
- Drug-Resistant Cell Models
- Cell Viability Assays
- Cell Proliferation Assays
- Cell Migration Assays
- Soft Agar Colony Formation Assay Service
- SRB Assay
- Cell Apoptosis Assays
- Cell Cycle Assays
- Cell Angiogenesis Assays
- DNA/RNA Extraction
- Cellular Phosphorylation Assays
- Stability Testing
- Sterility Testing
- Endotoxin Detection and Removal
- Phagocytosis Assays
- Ferroptosis Assay
- Cell-Based Screening and Profiling Services
- 3D-Based Services
- Custom Cell Services
- Cell-based LNP Evaluation
-
Stem Cell Research
- iPSC Generation
- iPSC Characterization
-
iPSC Differentiation
- Neural Stem Cells Differentiation Service from iPSC
- Astrocyte Differentiation Service from iPSC
- Retinal Pigment Epithelium (RPE) Differentiation Service from iPSC
- Cardiomyocyte Differentiation Service from iPSC
- T Cell, NK Cell Differentiation Service from iPSC
- Hepatocyte Differentiation Service from iPSC
- Beta Cell Differentiation Service from iPSC
- Brain Organoid Differentiation Service from iPSC
- Cardiac Organoid Differentiation Service from iPSC
- Kidney Organoid Differentiation Service from iPSC
- GABAnergic Neuron Differentiation Service from iPSC
- Undifferentiated iPSC Detection
- iPSC Gene Editing
- iPSC Expanding Service
- MSC Services
- Stem Cell Assay Development and Screening
- Cell Immortalization
-
Molecular Biology Solutions
- Chromosome & Genomic Analysis
-
Cytogenetics & Molecular Cytogenetics Analysis
- Fluorescent In Situ Hybridization (FISH)
- In Situ Hybridization (ISH) & RNAscope
- ImmunoFISH (FISH+IHC)
- I-FISH
- Splice Variant Analysis (FISH)
- RNA FISH in Plant
- mtRNA Analysis (FISH)
- Digital ISH Image Quantification and Statistical Analysis
- Telomere Length Analysis (Q-FISH)
- Telomere Length Analysis (qPCR assay)
- Droplet Digital PCR (ddPCR)
- QuantiGene Plex Assay
- Probe Development & Quality Control
- ISH/FISH Analysis for Therapeutic R&D
- Cell Line Characterization
- Pathogen & Microbial Analysis
- Histology Services
- Exosome Research Services
- Drug Metabolism and Pharmacokinetics (DMPK)
-
Safety Evaluation Services
- High-Content Cytotoxicity Screening
- High-Throughput Toxicity Screening
- In Vitro Cardiotoxicity
- In Vitro Genotoxicity
- Hepatotoxicity
- In Vitro Neurotoxicity
- In Vitro Nephrotoxicity
- In Vitro Dermal Toxicology
- Ocular Toxicity
- In Vitro Cytotoxicity
- Endocrine Disruption Screening Assay
- In Vivo Toxicity Study