CYP450 Time-Dependent Inhibition (TDI) Assay
- Service Details
- Features
- FAQ
- Explore Other Options
Time-dependent inhibition (TDI) of CYP450 enzymes is a major cause of clinically significant drug-drug interactions (DDIs). Unlike reversible CYP inhibition, TDI often involves metabolic activation and irreversible enzyme inactivation. Compounds forming reactive metabolites may permanently suppress CYP activity, leading to prolonged enzyme suppression and unexpected drug accumulation.
Understanding which specific CYP isoform is affected through reaction phenotyping studies is critical for mechanistic DDI assessment. In early development, TDI testing is frequently conducted alongside metabolic stability assays to provide a comprehensive evaluation of metabolic risk.
Regulatory agencies including FDA and EMA require TDI assessment during drug development to evaluate DDI risk. Creative Bioarray provides comprehensive in vitro CYP450 TDI assays using human liver microsomes to support early screening and regulatory submission.
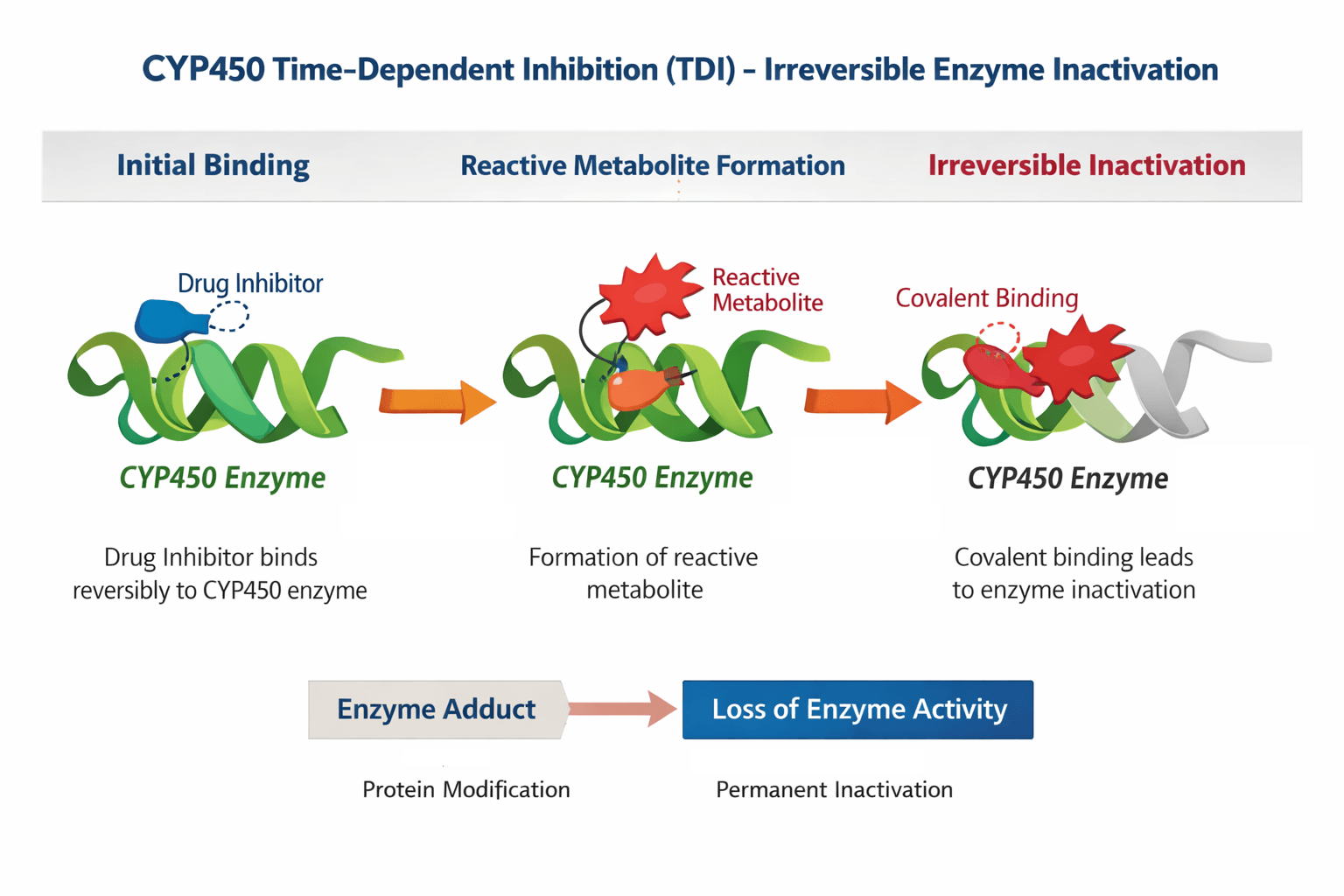 Fig. 1. Mechanism of CYP450 Time-Dependent Inhibition (TDI).
Fig. 1. Mechanism of CYP450 Time-Dependent Inhibition (TDI).
When Should You Perform a TDI Study?
You should consider TDI testing if:
- Your compound shows CYP inhibition in reversible screening
- Structural alerts suggest reactive metabolite formation
- CYP3A4 or CYP2D6 involvement is expected
- Regulatory DDI risk assessment is required for IND
- You need KI and kinact parameters for PBPK modeling
Creative Bioarray Offers Comprehensive CYP450 Time-Dependent Inhibition (TDI) Assay Services:
In the early drug screening phase, Creative Bioarray provides high-throughput, cost-effective TDI testings such as IC50 shift. In later stages, comprehensive enzyme kinetic studies can be conducted to obtain parameters like KI and Kinact for clinical implications.
IC50 shift assay (Early Screening) :
- Pre-incubation: Conduct pre-incubation at 37°C with or without NADPH, alongside a control group without inhibitor. A 30-minute pre-incubation time is typically used.
- Measurement phase: At each time point, add substrates to the pre-incubation system and commence enzyme activity measurements. Assess enzyme activity by determining substrate consumption via LC-MS/MS.
- Data processing: Calculate the IC50 Shift fold by comparing IC50 values with and without NADPH. A fold greater than a defined threshold (usually 1.5 or 2) indicates time-dependent inhibition.
| Parameter | Details |
|---|---|
| Matrix | Pooled human liver microsomes |
| CYP Isoforms | CYP1A2, 2A6, 2B6, 2C8, 2C9, 2C19, 2D6, 2E1, 3A4 |
| Compound Requirement | 5-10 mg of dry compound or 100 µL of 10 mM stock DMSO solution |
| Deliverables | IC50, Shifted IC50, and report. |
Service Features
Creative Bioarray offers cell immortalization service, including but NOT limited to:
- Advanced technology platform.
- Comprehensive service offerings.
- Expert technical team.
- Rigorous quality control.
- Flexible collaboration models.
FAQ
1: What is CYP450 time-dependent inhibition?
CYP450 time-dependent inhibition refers to certain drugs exhibiting marked inhibition effects after a period of combining with CYP450 enzymes.
2: Why is CYP450 TDI testing necessary?
CYP450 TDI testing predicts potential drug interaction risks, providing crucial insights for the research and optimization of drug metabolic properties.
3: What do I need to provide for testing?
Adequate samples of test compounds and associated chemical and physical property information are required.
4: Which types of CYP450 enzymes can be tested?
We can test a wide array of CYP450 enzymes, including but not limited to common types such as CYP1A2, CYP2A6, CYP2B6, CYP2C8, CYP2C9, CYP2C19, CYP2D6, CYP2E1, and CYP3A4.
5: Which drugs are suitable for CYP450 TDI testing?
TDI testing is applicable to all drug candidates needing evaluation of potential drug interaction risks.
Explore Other Options