Parallel Artificial Membrane Permeability Assay (PAMPA)
- Service Details
- Features
- FAQ
Membrane permeability and intestinal absorption are important properties to understand early in drug development to minimize late-stage attrition. Our PAMPA service offers a rapid, low-cost, high throughput in vitro model to assess passive permeability before moving into more sophisticated cell-based or in vivo models.
Using a lipid-infused artificial membrane system, PAMPA enables quantitative assessment of compound transport from a donor to an acceptor compartment, offering a reliable early ADME screening tool for compound prioritization. Since there are no living cells involved in the PAMPA assay, you get clean, reproducible data you can use to make go/no-go decisions about your compounds during hit-to-lead and lead optimization.
When Should You Choose PAMPA?
PAMPA is particularly well suited for projects that require rapid decision-making and efficient resource allocation, including:
- Early-stage screening of large compound libraries
- Prioritization of hits before investing in Caco-2 or MDCK assays
- Evaluation of passive transcellular permeability without transporter interference
- Initial assessment of oral absorption potential or blood–brain barrier (BBB) penetration
For compounds where active transport or efflux mechanisms are expected to play a major role, PAMPA is best used in combination with cell-based permeability models.
Our PAMPA Methods
We offer specialized PAMPA models designed to simulate key biological barriers relevant to drug absorption and distribution:

GIT-PAMPA: It is used to evaluate the permeability of drugs in the gastrointestinal tract and simulate the intestinal environment.

BBB-PAMPA: This method assesses the permeability of compounds across the blood-brain barrier.

Skin-PAMPA: Specializes in evaluating the permeation of compounds through the skin barrier.
Workflow
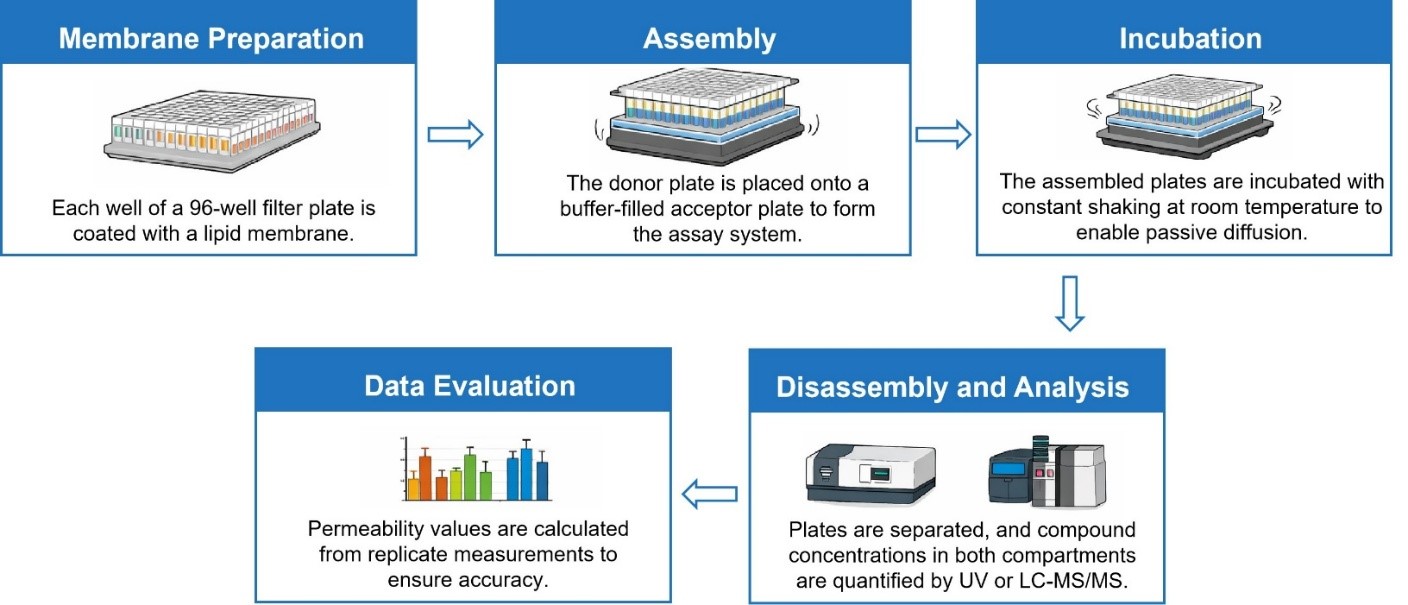
| PAMPA | Details |
| Test Article Concentration | 10 μM |
| Number of Replicates | 3 |
| Membrane Composition | Tri-layers of Phospholipids (Pre-coated) |
| Incubation Time | 4 hours |
| Test Article Requirements | 50 µL of 10 mM DMSO solution Brutto formulas are required. |
| Analysis Method | LC-MS/MS quantification |
| Data Delivery | Permeability (x10-6 cm/s). Full study report is provided. |
| Positive Control | Propranolol (high permeability) |
| Negative Control | Atenolol (low permeability) |
Why Work with Us?

Proven Expertise in Permeability Testing
Our scientists bring extensive experience in in vitro ADME screening, ensuring accurate data generation and interpretation.

Reliable and Decision-Ready Data
Standardized protocols and strict quality controls deliver reproducible results suitable for internal decision-making pipelines.

Flexible and Customizable Study Design
Assay conditions, analytical methods, and reporting formats can be tailored to meet specific project needs.

Technical Consultation Support
Our experts help you determine whether PAMPA is the right model for your compound and how to integrate results with downstream assays.
FAQ
Q1: How should PAMPA data be interpreted?
PAMPA evaluates drug permeability through artificial membranes by analyzing passive diffusion which serves as the main route through which drugs are absorbed in the gastrointestinal tract as well as across the blood-brain barrier and cellular membranes. The classification of compounds into either low permeability (Pe < 1.5 x 10-6 cm/s) or high permeability (Pe > 1.5 x 10-6 cm/s) assists researchers in selecting potential candidates for additional development processes.
Q2: Is PAMPA applicable to all types of compounds?
PAMPA provides excellent evaluation of passive transport attributes but lacks the ability to measure active transport processes. To obtain complete absorption data for compounds, researchers should combine PAMPA testing with cell-based methods such as Caco-2 assays.
Q3: What are the differences between PAMPA and other permeability testing methods?
The primary distinctions between PAMPA and permeability testing methods such as Caco-2 and MDCK cell models lie in the type of model they use and their operational complexity.
- Cell-free model: PAMPA employs synthetic membranes in its cell-free design while Caco-2 and MDCK models operate using live cells. The use of PAMPA removes any influence of cellular differences and metabolic processes when measuring permeability.
- Simplified operation: PAMPA provides a simpler operation compared to cell-based models because it eliminates the need for cell culture which streamlines experimentation and decreases time requirements.
- High-throughput screening: PAMPA's simplified operational process makes it exceptionally suitable for high-throughput screening which allows rapid permeability evaluation of compounds during early drug development phases.
- Cost-effectiveness: PAMPA provides a more cost-effective permeability testing option than cell models such as Caco-2 because it eliminates the need for cell culture.
- Data comparability: Caco-2 and MDCK models potentially deliver data that better represents physiological conditions for complex transmembrane permeability characterization because they reproduce biological barriers more accurately than PAMPA which provides useful permeability data.
Quotation and ordering
If you have further inquiries or need additional assistance, please contact our expert team.
Reference
- Ponmozhi J, Dhinakaran S, et al. Development of Skin-On-A-Chip Platforms for Different Utilizations: Factors to Be Considered. Micromachines. 2021; 12(3):294.
Explore Other Options