Biomarkers vs. Functional Assays: Closing the Preclinical Gap
In the high-stakes world of preclinical drug development, there is a recurring nightmare that keeps R&D heads awake at night: the "successful" failure.
It looks like this: Your lead candidate is a star performer in early testing. The Biomarkers are perfect-target engagement is hitting 90%, protein inhibition is crystal clear on a Western Blot, and the PK profile is exactly where it should be. On paper, you have a winner.
But then, you move into Functional Assays, and the reality is a cold shower. The tumor cells keep proliferating. The inflammatory cytokine storm doesn't subside. The biological "needle" doesn't move. Your drug is like a key that fits perfectly into the lock but refuses to turn.
At our CRO, we call this the "Two-Way Mirror" paradox. Biomarkers show you what the drug is doing to the target, but Functional Assays show you what the drug is doing for the patient. If you can't align the two, you aren't developing a medicine; you're just documenting a molecular interaction.
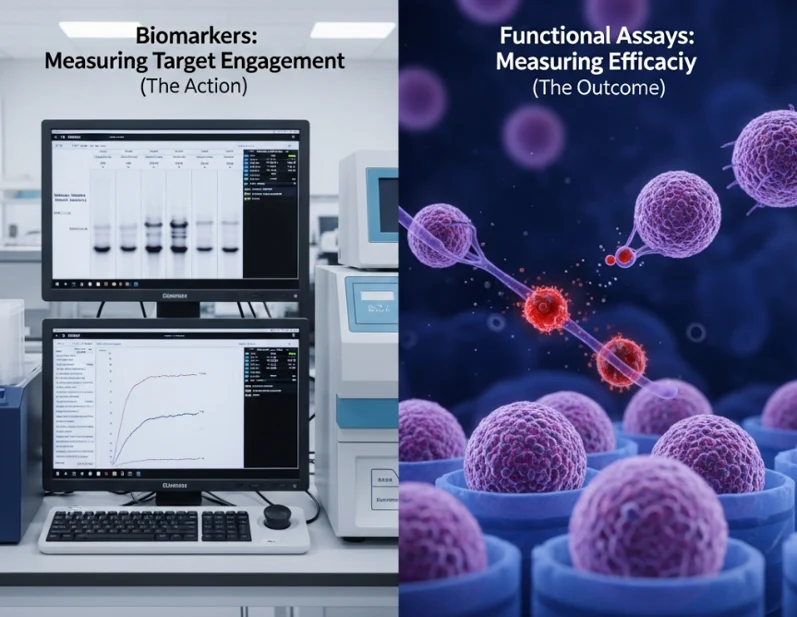
Biomarkers: The Navigators (But Not the Destination)
Biomarkers are the eyes of your project. In the current regulatory landscape, having robust Pharmacodynamic (PD) data is non-negotiable for an IND filing. We use them to prove "Target Engagement"-the essential first step of showing that your molecule actually reached its destination.
With today's ultra-sensitive detection platforms, we can measure picoscale changes in protein expression or genetic shifts. This gives us immense confidence. However, a biomarker is often just a proxy. It tells you the door was unlocked, but it doesn't guarantee that anyone actually walked through it. Relying solely on biomarkers is like checking your GPS and seeing you've arrived at the restaurant, without checking to see if the kitchen is actually open.
Functional Assays: The Ultimate Reality Check
If biomarkers are the map, functional assays are the terrain. While biomarkers measure "action," functional assays measure "outcome."
The regulatory tide is shifting. With the recent FDA Modernization Act 2.0, there is a massive push toward New Alternative Methods (NAMs). Regulators and investors are no longer satisfied with seeing a "shrunken band" on a gel; they want to see functional reversal of disease.
Whether it's a complex cell-killing assay or a GPCR signaling test, these experiments tell us if the molecular binding actually translates into a physiological change. This is the "End-State" data that de-risks your transition into Phase I clinical trials.
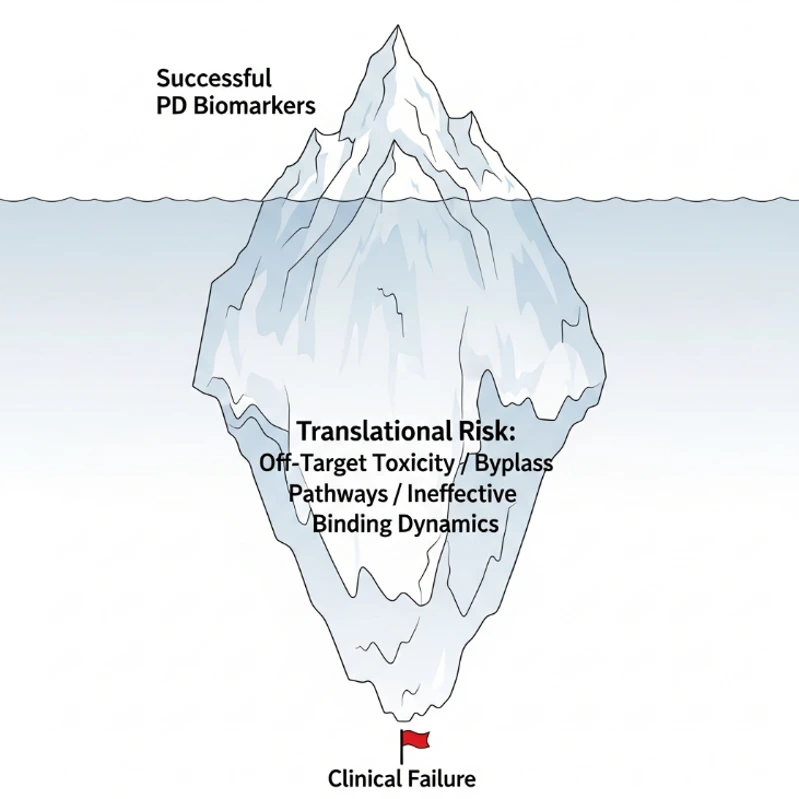
Why the Data Disconnects: The Three Great Gaps
Why do biomarkers and functional results often tell two different stories? In our years of troubleshooting for global biopharma, we've identified three primary culprits:
The "Bypass" Problem (Cellular Compensation)
Biological systems are incredibly resilient. They aren't linear tracks; they are vast, redundant networks. You might successfully shut down Target A (Biomarker: Success!), but the cell is smart-it immediately reroutes traffic through "Pathway B" to keep growing. The result? Your biomarker says the target is dead, but the functional assay shows the cell is very much alive.
"Sitting" vs. "Acting" (Binding Dynamics)
Not all binding is created equal. A molecule might have high affinity-it "sits" on the receptor-but if the residence time is too short or it binds to an epitope that doesn't trigger a conformational change, nothing happens downstream. This is "silent binding." It looks great in a binding assay but produces zero functional response.
The Microenvironment "Mask"
Most biomarkers are measured in peripheral blood or simple cell media. But in the real world-especially in oncology or autoimmune disease-the Tumor Microenvironment (TME) is a hostile place. Physical barriers, pH changes, and interstitial pressure can stop a drug that "binds" perfectly in a test tube from ever performing its "function" in a solid tissue.
The 2026 Strategy: Moving Beyond Linear R&D
To survive the "Valley of Death" between preclinical and clinical stages, you need to stop viewing Biomarkers and Functional Assays as separate checkboxes. You need an Integrated Strategy:
- Correlation, Not Sequence: Don't wait for "perfect" biomarkers to start functional testing. Run them in parallel to establish a "Binding-to-Function" ratio early.
- Human-Centric Models: Move away from immortalized cell lines as soon as possible. Use Patient-Derived Organoids (PDOs) and primary human cells. If the function doesn't work in a human-relevant model, it won't work in a human patient.
- Mechanism-of-Action (MOA) Deep Dives: Use AI and transcriptomics to see the "whole picture." If a biomarker moves but the function doesn't, find out why the cell is compensating before you spend millions on a clinical trial.
Conclusion
In the race to the clinic, data that is merely "positive" isn't enough. It has to be predictive.
Biomarkers give you the confidence to move forward, but Functional Assays give you the evidence that you're headed in the right direction. As your preclinical partner, our job is to look behind the "mirror"-to ensure that when your biomarkers say "Yes," your functional outcomes say "Yes" too.
Don't let your drug be a "successful" failure. Let's find the data that actually moves the needle.
Creative Bioarray Relevant Recommendations
| Products & Services | Description |
|---|---|
| PK/PD Biomarker Analysis | Creative Bioarray offers comprehensive PK/PD biomarker analysis to optimize dosing, predict efficacy, ensure safety, and accelerate drug development. |