Mesenchymal Stem Cells (MSCs) Services
In recent years, due to the high growth potential and multipotent differentiation ability of mesenchymal stem cells (MSCs), their application in cell therapy has received extensive attention. As a trusted research partner in the field of biotherapy, Creative Bioarray provides advanced and experienced cell therapy research services covering all stages of MSC development, including isolation, expansion, characterization and differentiation.
Therapeutic potential of MSCs
- MSCs have the capacity of differentiating into non-mesenchymal lineages, including neuronal cells, endothelial cells, astrocytes, cardiomyocytes and so on.
- MSCs provide great therapeutic benefits by secreting soluble factors to induce an immunomodulatory environment.
- MSCs show ability to migrate to injury sites and tumor microenvironments, thus can be used as a carrier for targeted therapy.
- MSCs are widely available and can be easily expanded ex vivo through different protocols.
MSC Isolation and Expansion service
MSCs come from different sources and can be isolated from various tissues, such as umbilical cord, bone marrow, cartilage, muscle, and adipose tissues. In addition, it is reported that fetal dermis, various tooth tissues, pancreas tissues, breast milk, menstrual blood and endometrium also contain MSCs. MSCs from different tissues have different characteristics, including expression of cell surface markers and differentiation abilities.
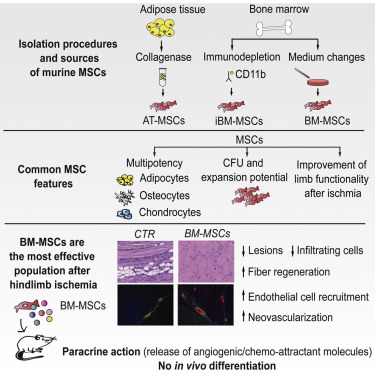
Creative Bioarray uses different protocols for MSCs isolation
- MSCs can be obtained by seeding bone marrow cells on tissues plastic dishes, and then washing away non adherent cells due to the adhesion property of MSCs.
- Extracting monocytes from bone marrow by density gradient centrifugation or enzymatic treatment of solid tissues with collagenase can be used to increase the yield of MSCs.
- Combined with fluorescence-activated cell sorting and magnetic beads, immunoseparation and immunodepletion technologies can improve the purity of the obtained MSCs depending on different surface markers.
MSC Characterization service
Since the population heterogeneity of MSC culture is caused by factors such as different tissue derivation, diversified isolation, and multiple growth conditions, it is necessary to define genera; guidelines for MSC preparation to characterize MSC populations. The International Society for Cell Therapy (ISCT) announced the basic characteristics and minimum standards of MSC in 2006. Since then, other markers have been recognized and widely accepted as reliable MSC surface markers, such as STRO-1, CD271, CD348, CD200 and CD105.
Creative Bioarray provides comprehensive characterization services for MSCs
- Immunophenotype analysis
- Tri-lineage Differentiation Potential analysis
- Immunomodulation analysis
- Short Tandem Repeat (STR) analysis
- Karyotype analysis
MSC Differentiation service
MSCs can differentiate into a variety of specialized cell types, including:
- Mesoderm: osteoblasts, chondrocytes, adipocytes, cardiomyocytes
- Ectoderm: neuronal cells
- Endoderm: hepatic, pancreatic, respiratory epithelial cells
Example of MSC Characterization Results:
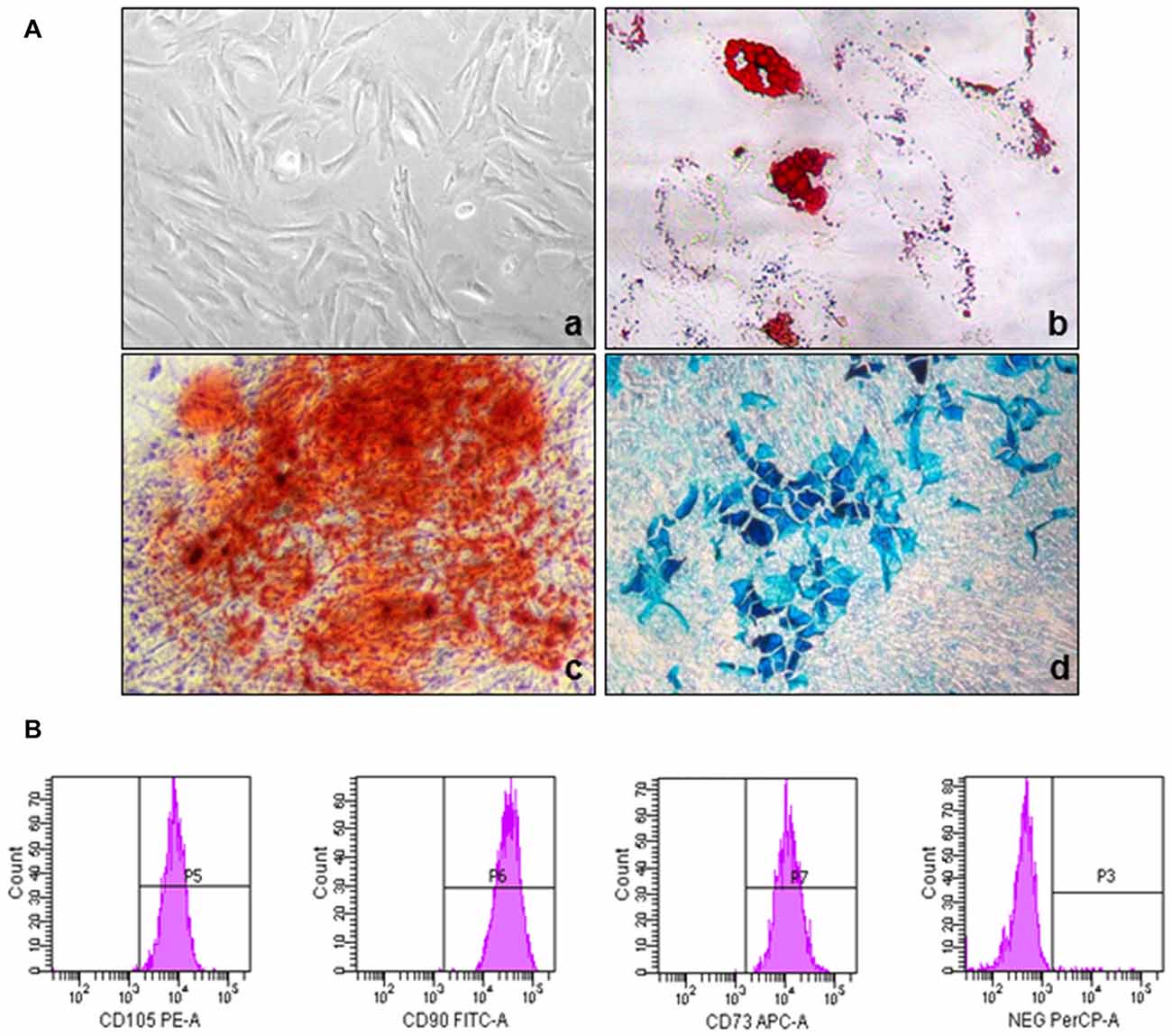 Fig 1: Phenotypic and functional characterization of MSCs. (A) The morphology of umbilical cord-derived MSC and their differentiation into adipogenic, chondrogenic, and osteogenic lineages. (B) Immunophenotypic characterization of UC-MSC by Flow Cytometry.
Fig 1: Phenotypic and functional characterization of MSCs. (A) The morphology of umbilical cord-derived MSC and their differentiation into adipogenic, chondrogenic, and osteogenic lineages. (B) Immunophenotypic characterization of UC-MSC by Flow Cytometry.
Creative Bioarray is an experienced and outstanding provider of Mesenchymal Stem Cells (MSCs)-related services. We are dedicated to providing quality data and comprehensive service for your scientific research, and we are pleased to use our extensive experience and advanced platform to offer the best service to satisfy each demand from our customers.
If you have any special need in Mesenchymal Stem Cells (MSCs) service, do not hesitate to contact us for this special service. Please let us know what you need and we will accommodate you. We are looking forward to working with you in the future.
Explore Other Options