Reaction Phenotyping vs. Metabolic Stability
In the early discovery process of drug development, many scientists get puzzled:
"We already did metabolic stability testing and passed. Do we still need reaction phenotyping analysis?"
What is the difference between CYP phenotyping and clearance studies anyways?"
The questions above are understandable as reaction phenotyping and metabolic stability seem like two overlapping tests that both study "clearance." However, the focus, scientific questions, and implementation timelines of these experiments are entirely different. Briefly,
Metabolic Stability tells you, "How fast is it cleared?"
Reaction Phenotyping tells you, "By whom (which enzyme) is it cleared?"
Knowing how and when to use these two tools properly can optimize your ADME research plan, speed up your compound screening process, and avoid pitfalls down the development road.
What is Reaction Phenotyping?
Reaction phenotyping aims to identify which enzymes are responsible for metabolizing the drug of interest. More specifically, reaction phenotyping drills down to identifying which isoforms of CYPs (CYP3A4, 2D6, 2C9…etc) are the major players. Key questions being asked:
- Who cleared the drug?
- Which CYP isoform is responsible for the majority of metabolism?
This research is the cornerstone of assessing Drug-Drug Interaction (DDI) risks. If a drug candidate's metabolism relies heavily on a single CYP enzyme that can be inhibited or induced (such as CYP3A4), severe clinical DDI events may occur when co-administered with strong inhibitors (like ketoconazole) or inducers (like rifampin).
Common Experimental Methods
- Recombinant human CYP enzymes (rhCYP): Drug of interest incubated with cells that overexpress only one CYP isoform at a time. The relative contribution of each enzyme is determined by comparing metabolism rates.
- Chemical inhibition: Specific chemical inhibitors that inhibit enzymes one at a time (quinidine-inhibits CYP2D6) are added into HLM incubations to determine how much drug metabolism is decreased.
- Correlation analysis: Metabolism is performed using a bank of liver microsome from different donors that have known enzyme levels. Correlation between enzyme levels and drug clearance rates are then determined.
Key Outputs and Interpretation
- Percentage contribution of CYP enzymes: Quantifies the relative contribution of each CYP isoform to total metabolism (e.g., CYP3A4 contributes 70%, CYP2C9 contributes 30%).
- Major metabolic enzyme identification: Clearly identifies the dominant enzyme driving the drug's metabolism.
- DDI risk indicators: Provides a basis for designing subsequent clinical DDI study protocols based on the characteristics of the major enzyme (e.g., whether it is polymorphic or highly susceptible to inhibition/induction).
What is Metabolic Stability?
Metabolic Stability (MS) is a test that determines how fast a compound is cleared in vitro (i.e. liver microsome or hepatocytes). Key questions being asked:
- How fast is it being cleared?
- Is the drug prone to fast hepatic clearance which will result in a short half-life and poor exposure in vivo?
This research serves as a "compass" for medicinal chemists during structural optimization, helping to quickly eliminate compounds with excessive metabolic rates and poor drug-like properties.
Common Experimental Systems
- Liver microsome: The most common system used to screen compounds. Contains all major phase I metabolic enzymes (CYPs…etc.), some phase II conjugating enzymes. It's cheap and highly parallelizable.
- Hepatocytes: System that more closely represents in vivo environment. Has intact cellular structure, full complement of metabolic enzymes, and transporters. Allows you to study phase I/II metabolism and cellular uptake simultaneously.
- S9 fraction: Contains both cytosolic and microsomal fraction in equal ratio. Great for studying phase I/II metabolism but doesn't have the cellular components.
Key Outputs and Interpretation
- Half-life (t1/2): How long it takes for the compound to decrease by half in the incubation system.
- Intrinsic Clearance (CLint): A theoretical clearance value calculated from parameters like half-life, used to predict in vivo hepatic clearance.
- Percentage of Remaining Parent Drug: The proportion of the original compound left unmetabolized after a fixed time point (e.g., 60 minutes), serving as a common metric for rapid screening.
When to Do Reaction Phenotyping vs Metabolic Stability?
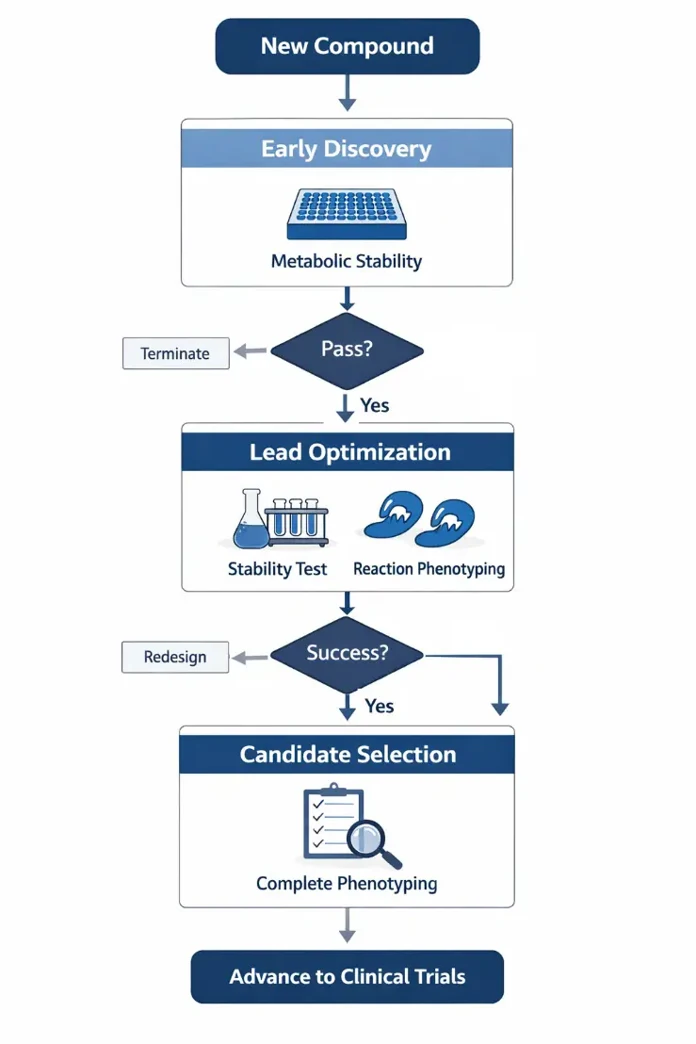
Early Discovery Stage
- Priority: Metabolic Stability Testing.
- Reasoning: At this stage, libraries are vast. The primary goal is to quickly weed out molecules with poor metabolic profiles. These tests are high-throughput and cost-effective, providing clear optimization directions for the chemistry team.
Lead Optimization to IND Filing
- Priority: Integrated Metabolic Stability + Reaction Phenotyping.
- Reasoning: Once a candidate meets stability requirements, "stable" does not mean "DDI-safe." A stable compound primarily metabolized by CYP3A4 may still carry high clinical DDI risks. Introducing reaction phenotyping here identifies potential DDI "traps" early, informing clinical development strategies and meeting regulatory requirements.
Clinical and Regulatory Considerations
- Reaction phenotyping data forms the foundation for clinical pharmacology plans, specifically deciding if and how to conduct clinical DDI studies per FDA/EMA guidelines.
- Metabolic stability data (especially from hepatocytes) continues to support modeling and simulation for human PK prediction and dose selection.
Common Misconceptions in ADME Studies
- "If metabolic stability is good, I don't need reaction phenotyping."
Fact: Good stability only means slow clearance. If that clearance path relies on a single enzyme, the DDI risk may actually be higher. Phenotyping reveals these "hidden risks" beneath a stable profile.
- "Reaction phenotyping can directly predict in vivo PK (e.g., half-life or AUC)."
Fact: Phenotyping identifies who the metabolizer is. In vivo PK is determined by a combination of stability, hepatic blood flow, protein binding, and transporters. It is a qualitative risk assessment tool rather than a quantitative PK predictor.
- "A single experimental system (like HLM) can solve all metabolic questions."
Fact: HLM is a great screening tool but cannot assess the full contribution of Phase II metabolism (like UGT) or cellular uptake. Comprehensive assessment requires multi-layered systems.
Frequently Asked Questions (FAQ)
Q1: If my compound doesn't have good metabolic stability, do I still need reaction phenotyping?
A: Low priority in the early phase. You should focus all your efforts on figuring out how to stabilize the compound. Once you've successfully stabilized the compound through optimization, running reaction phenotyping will be crucial to ensure you didn't trade away one problem for another.
Q2: Can I use metabolic stability data to predict drug-drug interaction?
A: Indirectly. Metabolic stability only addresses rate of clearance. DDI is concerned with WHICH enzyme got inhibited. A compound with a slow rate of metabolism but depends on a single enzyme for clearance (CYP3A4) will have a higher risk of DDI than a compound with a fast rate of metabolism cleared by multiple pathways.
Q3: Do I need to run both tests before IND submission?
A: Both should be critical pieces of information to justify your decision. Metabolic Stability supports PK prediction and species selection. Reaction Phenotyping is what the regulators want to see in terms of assessing clinical DDI risk.
Q4: Can I run both assays in parallel to save time?
A: Absolutely! That is not necessarily the recommended approach but it will not hurt to run them together. The strategy recommended here is staggered: screen/optimize with metabolic stability and run reaction phenotyping on select compounds that passed your MS cutoff values. For precious leads in late-stage optimization, running them in parallel can save you time.
Creative Bioarray Relevant Recommendations
| Products & Services | Description |
|---|---|
| CYP and UGT Reaction Phenotyping Assay | Creative Bioarray helps provide CYP and UGT reaction phenotyping assay, with years of experience and excellent scientific team to provide you with quality service. |
| Metabolic stability & identification | Comprehensive metabolic stability and metabolite identification services to de-risk drug development - leveraging physiologically-relevant models and cutting-edge LC-MS/MS technology for actionable ADME insights. |
| Drug-Drug Interaction | Creative Bioarray provides a range of high-quality drug-drug interaction services to meet FDA and EMA guidance. |