Cytochrome P450 Induction Assay
- Service Details
- Features
- FAQ
- Explore Other Options
Our CYP450 induction assay helps you quickly evaluate how your drug candidates affect liver metabolism and drug-drug interactions. Gain insights to optimize drug safety, dosing, and efficacy.
Cytochrome P450 (CYP450) enzymes are a class of enzymes widely involved in drug metabolism, particularly in the liver where they catalyze the oxidation of numerous drugs. Induction of CYP450 enzymes occurs primarily through nuclear receptors, such as the pregnane X receptor (PXR), the constitutive androstane receptor (CAR), and the aryl hydrocarbon receptor (AhR). By binding to ligands, such receptors can heterodimerize and diffuse to the nucleus, which in turn targets the regulatory sites of CYP enzyme genes and enhances the enzymes' gene expression. CYP450 induction is an important consideration in drug development and clinical application because it can significantly affect the pharmacokinetics and efficacy of drugs.
A drug that induces CYP can reduce the concentration of other drugs sharing the same metabolism pathway and thus reduce their efficacy or promote metabolite toxicity. Thus, to accurately forecast the likelihood of drug-drug interactions, it is essential that a correct assessment of the risk of CYP induction be made in the initial stages of drug development. Such predictions can avoid undesirable drug interactions and enhance drug safety and effectiveness.
Why cytochrome P450 induction assay?
- Provides insights into drug metabolism to optimize drug design, bioavailability, and efficacy.
- Helps predict how CYP450 enzymes affect drug levels, reducing risks of toxicity or reduced efficacy.
- Assesses safety, efficacy, and potential drug-drug interactions during development.
- Supports clinical decision-making by informing dosing and avoiding harmful drug combinations.
Assay Details
Workflow
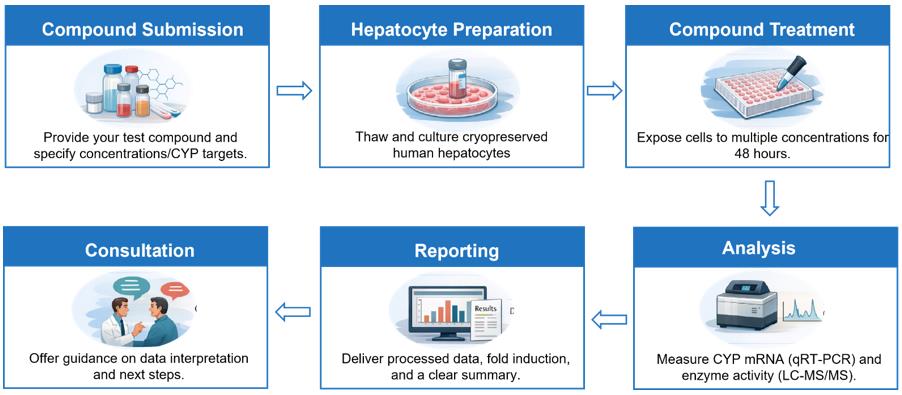
Protocol
| Test System | Cryopreserved human hepatocytes (3 donors) |
| Test Article Concentration | 5 concentrations (e.g. 0.1, 0.33, 1, 3.3, 10 µM, or concentrations of your choice), in triplicate |
| CYP Isoforms | CYP1A2, CYP2B6, and CYP3A4/5. For other CYPs, please contact us for more information |
| Number of Replicates | 3 |
| Negative Control | Vehicle (0.1% DMSO) |
| Positive Control | Omeprazole for CYP1A2 Phenobarbital for CYP2B6 Rifampicin for CYP3A4, CYP2C8, CYP2C9, CYP2C19 |
| Exposure Period | 48 h (Medium changed every 24 h with freshly diluted test articles) |
| Probe Substrates for Catalytic Activity | Phenacetin for CYP1A2 Bupropion for CYP2B6 Testosterone or Midazolam for CYP3A4 Amodiaquine for CYP2C8 Diclofenac for CYP2C9 S-mephenytoin for CYP2C19 |
| Analysis Method | qRT-PCR for relative mRNA expression levels. LC-MS/MS quantification of probe substrate metabolites: Acetaminophen: CYP1A2 Hydroxybupropion: CYP2B6 6β-Hydroxytestosterone or 1'-OH midazolam: CYP3A4 N-desethylamodiaqune: CYP2C8 4'-OH diclofenac: CYP2C9 4'-OH mephenytoin: CYP2C19 |
| Data Delivery | Emax, EC50, CYPs activity, mRNA levels of CYPs, fold induction relative to the positive control, Raw Data, Report |
| Test Article Requirements | 5-10 mg solid compound or 100 μL of a 10 mM stock solution is required, with the actual amount depending on the highest concentration. |
Our CYP450 Induction Assay Service Offers Several Distinct Advantages
Reliable Data
We use cryopreserved human hepatocytes and standardized protocols to generate consistent and reproducible results.

Comprehensive Testing
Assess major CYP isoforms (CYP1A2, CYP2B6, CYP3A4/5) with both enzyme activity and mRNA expression endpoints.
Flexible & Customizable
Test concentrations and CYP targets can be tailored to fit specific project needs.

Expert Support
Our experienced scientists provide guidance on study design, data interpretation, and troubleshooting.

Timely Reporting
Results are delivered promptly, including raw data, fold induction, EC50/Emax, and a clear summary report.
FAQ
1. What is the cytochrome P450 induction process?
The two main mechanisms for CYP450 (CYP450, or CYP) induction are stabilization of mRNAs (or enzymes) and transcription gene activation. Nuclear receptors (NRs) function as transcription factors, and they regulate the activation of transcribed genes. These nuclear receptors are the aryl hydrocarbon receptor (AhR), androstane receptor (CAR, also known as the constitutive androstane receptor), and pregnane X receptor (PXR). Such nuclear receptors get activated by binding to ligands and being moved from the cell to the nucleus. Nuclear receptors can heterodimerize with other proteins in the nucleus (retinoid X receptor RXR or transporter protein ARNT), which bind to target genes (CYPase genes) and drive expression of CYPases. Moreover, over the past few years, microRNAs have also been shown to post-transcribe CYP enzymes.
2. What are the types of CYP450 induction assays?
The most common cytochrome P450 induction assays are CYP1A2, CYP2B6, CYP2C8, CYP2C9, CYP2C19 and CYP3A4. We typically recommend CYP1A2, CYP2B6, and CYP3A4 as the primary target because they are activated by various nuclear receptors. Since CYP3A4 and CYP2C are induced by the same process, i.e., they both require activation of the PXR, we do not test whether CYP2C has been induced if no CYP3A4 has been induced by NCE and vice versa.
3. Why evaluate both activity and mRNA expression?
Endpoints in enzyme induction studies can be determined by measuring mRNA levels and/or enzyme activity levels using specific probe substrates. Enzyme inhibition might cause "false negatives", affecting enzyme activity assessments. In contrast, mRNA testing is not affected by this issue, providing an advantage. However, if a new chemical entity (NCE) prevents enzyme protein degradation, enzyme activity should also be considered as an evaluation criterion.
4. What are evaluation criteria for cytochrome P450 induction assay?
The fold-change method is generally used to assess induction effects. A potential induction is indicated if the fold-change in CYP enzyme mRNA, compared to solvent control, is ≥2 times at expected liver concentration and shows a concentration-dependent increase. If the mRNA fold-change is<2 but="" the="" increase="" is="">20% of that of the positive control drug, the possibility of enzyme induction cannot be ruled out and requires further testing for confirmation.
Quotation and Ordering
If you have any special needs or questions regarding our services, please feel free to contact us. We look forward to cooperating with you in the future.
Explore Other Options