HEK 293
Cat.No.: CSC-6980J
Species: Homo sapiens (Human)
Source: Kidney
Morphology: Adherent fibroblastoid cells growing as monolayer
Culture Properties: Adherent
- Specification
- Background
- Scientific Data
- Q & A
- Customer Review
The Human Embryonic Kidney 293 (HEK293) cell line stands as one of the most widely adopted and thoroughly validated mammalian expression systems in modern biomedical research. Originally established in 1973 by Alex van der Eb and Frank Graham at Leiden University through stable transfection of normal human fetal kidney cells with sheared adenovirus type 5 DNA, HEK293 cells have since become an indispensable platform across academic, biotechnology, and biopharmaceutical settings - second only to HeLa cells in global research utilization.
A defining strength of HEK293 cells lies in their exceptional transfection efficiency, readily accepting foreign genetic material via a broad spectrum of chemical, physical, and viral delivery methods, significantly reducing experimental turnaround compared to alternative cell lines such as CHO. As a human-origin cell line, HEK293 cells are uniquely capable of performing complex human-like post-translational modifications (PTMs) - including glycosylation, phosphorylation, and γ-carboxylation - ensuring that recombinant proteins are produced with authentic biological activity and structural fidelity that non-human systems fundamentally cannot replicate.
HEK293 cells exhibit rapid proliferation, robust adaptability to both adherent and suspension culture formats, and proven compatibility with serum-free media, enabling scalable protein production under GMP-aligned conditions. Their consistent, batch-to-batch reproducibility makes them an ideal model for receptor signaling studies, viral vector manufacturing, drug screening, toxicology assessment, and the production of therapeutic proteins.
Furthermore, the versatile HEK293 cell family - including the widely used HEK293T variant expressing the SV40 Large T antigen for enhanced episomal replication - offers researchers flexible, fit-for-purpose solutions across transient and stable expression paradigms.
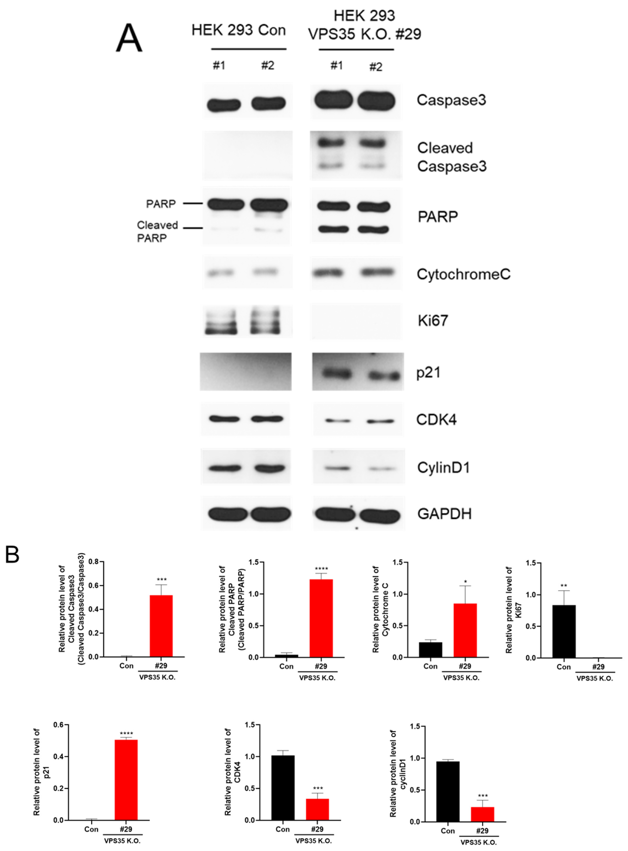
VPS35 Deficiency Markedly Reduces the Proliferation of HEK293 Cells
The retromer protein complex is involved in various physiological processes, especially endosomal trafficking, and its dysregulation has been linked to Alzheimer's disease and Parkinson's disease, as well as VPS35 knockout (KO), causing early embryonic lethality.
To investigate the effects of VPS35 loss, we used CRISPR/Cas9 to generate VPS35 KO human embryonic kidney 293 (HEK293) cells. We analyzed changes in retromer component expression, cell proliferation, apoptosis, and mitochondrial dynamics using Western blotting, terminal deoxynucleotidyl transferase dUTP nick end labeling (TUNEL) assay, and confocal microscopy.
The results showed that VPS35 KO significantly reduced cell proliferation and decreased expression of VPS29 and VPS26, both essential for retromer complex assembly. Compared to control cells, KO cells exhibited elevated levels of cleaved caspase-3, poly(ADP-ribose) polymerase, cytochrome C, and p21, while the expression of Ki-67, CDK4, and cyclin D was reduced. Additionally, VPS35 deletion also promoted mitochondrial fragmentation, associated with increased expression of mitochondrial fission-related proteins. These findings suggest that VPS35 plays a critical role in cell growth and survival by modulating apoptosis, mitochondrial dynamics, and cell cycle progression.
Ask a Question
Write your own review
- Adipose Tissue-Derived Stem Cells
- Human Neurons
- Mouse Probe
- Whole Chromosome Painting Probes
- Hepatic Cells
- Renal Cells
- In Vitro ADME Kits
- Tissue Microarray
- Tissue Blocks
- Tissue Sections
- FFPE Cell Pellet
- Probe
- Centromere Probes
- Telomere Probes
- Satellite Enumeration Probes
- Subtelomere Specific Probes
- Bacterial Probes
- ISH/FISH Probes
- Exosome Isolation Kit
- Human Adult Stem Cells
- Mouse Stem Cells
- iPSCs
- Mouse Embryonic Stem Cells
- iPSC Differentiation Kits
- Mesenchymal Stem Cells
- Immortalized Human Cells
- Immortalized Murine Cells
- Cell Immortalization Kit
- Adipose Cells
- Cardiac Cells
- Dermal Cells
- Epidermal Cells
- Peripheral Blood Mononuclear Cells
- Umbilical Cord Cells
- Monkey Primary Cells
- Mouse Primary Cells
- Breast Tumor Cells
- Colorectal Tumor Cells
- Esophageal Tumor Cells
- Lung Tumor Cells
- Leukemia/Lymphoma/Myeloma Cells
- Ovarian Tumor Cells
- Pancreatic Tumor Cells
- Mouse Tumor Cells





