What Are Biomarkers in Drug Discovery?
What Are Biomarkers?
Bio-markers, also known as biological markers, are objectively measured and evaluated indicators of normal biological processes, pathogenic processes, or pharmacologic responses to a therapeutic intervention.
Examples of biomarkers can include things found in biological samples such as blood or urine but can also include imaging. Biomarkers can also indicate:
- Cell activity or molecule secretion (cytokines)
- Genetic changes (gene expression signature)
- Protein levels or modifications
- Physiology or imaging changes
Some of the earliest review articles published about drug development categorized biomarkers as "devices that translate biological information into numeric data".
In drug development, numeric data is changed we measure to help inform future decisions during the drug discovery and development process. Notice there is overlap in how people may define each of these biomarker types.
Typically, biomarkers are grouped into categories based on their function. There are also subcategories that describe the type of molecule that is being measured. Learning about how each of these biomarker types are used will allow you to better design and use biomarkers in your drug discovery program.
Types of Biomarkers in Drug Development
Over the past few years, you will notice that most review articles define biomarkers into similar categories. The categories define the role the biomarker plays in drug development. The table below includes examples of each biomarker type along with examples that have been well characterized in preclinical and clinical studies.
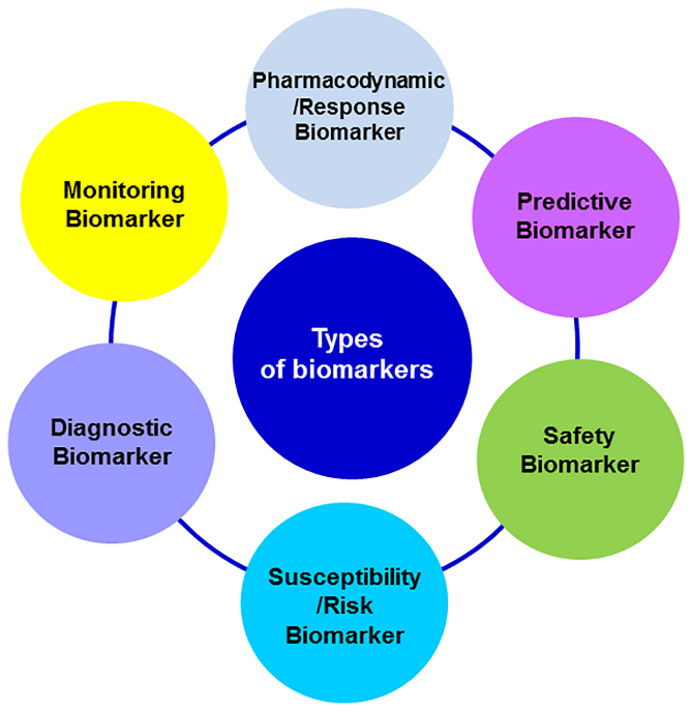
Diagnostic Biomarkers
Diagnostic biomarkers are used to detect or confirm the presence of a disease. They are essential in both clinical diagnosis and in validating disease models during preclinical research.
Examples:
- Prostate-specific antigen (PSA) for prostate cancer detection
- HbA1c levels for diagnosing and monitoring diabetes
- Amyloid-β (Aβ) accumulation detected by PET imaging in Alzheimer's disease
Identifying disease in preclinical models is analogous to diagnostics. For example, researchers may verify they have successfully induced inflammation by measuring the levels of inflammatory cytokines like IL-6 or TNF-α.
Prognostic Biomarkers
Prognostic biomarkers offer insight into the likely course of a disease, irrespective of treatment. These are useful for establishing study endpoints and disease stratification.
Examples:
- Ki-67 proliferation index indicating tumor aggressiveness
- C-reactive protein (CRP) levels associated with inflammation severity and cardiovascular risk
- Tumor mutational burden (TMB) as an indicator of cancer progression potential
Tumor size, degree of fibrosis, or neuron breakdown could be used as prognostic biomarkers in a preclinical study to help determine how long the study should run.
Predictive Biomarkers
Predictive biomarkers are used to identify whether a patient will respond to a given drug therapy. They are crucial for precision medicine applications.
Examples:
- HER2 overexpression predicting response to trastuzumab in breast cancer
- EGFR mutations predicting response to tyrosine kinase inhibitors (e.g., gefitinib)
- PD-L1 expression guiding immunotherapy eligibility
Examples of predictive biomarkers are used frequently in clinical trials these days, especially for oncology indications. Predictive biomarkers could also be used in research studies with animals or cells to determine if your drug is effective in your model of interest.
Pharmacodynamic (PD) Biomarkers
PD markers are indicative of drug response. They are directly related to the target or disease mechanism you are interested in. These biomarkers frequently become the focus for demonstrating that a drug hits its intended target and achieves the desired effect.
Examples:
- Phosphorylated ERK (p-ERK) levels as a readout of MAPK pathway inhibition
- Cytokine reduction (e.g., IL-6, TNF-α) following anti-inflammatory treatment
- Gene expression changes measured by qPCR or RNA-seq after drug administration
PD biomarkers can be classified into:
- Proximal biomarkers (direct target engagement, e.g., receptor occupancy)
- Distal biomarkers (downstream biological effects)
In preclinical CRO studies, PD biomarkers are routinely measured using ELISA, multiplex assays, flow cytometry, or transcriptomic profiling.
Safety Biomarkers
Safety biomarkers are used to detect or predict adverse effects and toxicity. They are crucial in both preclinical toxicology studies and early-phase clinical trials.
Examples:
- Alanine aminotransferase (ALT) and aspartate aminotransferase (AST) for liver toxicity
- Cardiac troponins (cTnI, cTnT) for cardiotoxicity
- Kidney injury molecule-1 (KIM-1) for renal toxicity
In preclinical studies, safety biomarkers enable early detection of organ-specific toxicity, improving risk assessment and reducing late-stage drug failure
What's Trending in Biomarkers?
The biomarker space is quickly changing. Some issues gaining a lot of study in the realm of biomarkers include:
Multi-omics Integration
Combining genomic, transcriptomic, proteomic, and metabolomic data together is another growing sector. Through multi-omics approaches, biological complexity can be understood at several levels of regulation which can lead to more specific and robust biomarkers.
Research has found that biomarkers can be used:
- In cancer drug discovery
- Neurodegenerative disease modeling
- In immunology and inflammatory disease research
These were just found by utilizing multi-omics data to develop the biomarkers.
AI and Machine Learning
AI-enabled biomarker discovery is becoming a revolutionary approach for early drug development. By utilizing machine learning algorithms, patterns can be uncovered in high-dimensional biological data to:
- Prioritized biomarkers
- Selecting features from omics datasets
- Predicting drug response through modeling
A recent review on translational drug development revealed that AI simplifies and accelerates the process of early biomarker discovery with less experimentation.
Liquid Biopsy and Non-invasive Biomarkers.
Biomarkers that can be derived from blood or other bodily fluids are also known as liquid biopsies. These are becoming more widely used as they can act as non-invasive biomarkers. Unlike biomarkers derived from tumor biopsies, they can continue to monitor disease that may change over time.
Spatial Biology and Single-Cell Technology
Emerging spatial transcriptomics and single-cell sequencing technologies enable remarkable resolution in finding cell-type-specific biomarkers, hence increasing disease mechanism comprehension at the microenvironment level.
Applications of Biomarkers in Drug Discovery and Clinical Trials
Biomarkers play a critical role in transforming clinical trial design and improving the efficiency of drug development pipelines.
Early Drug Discovery
In early stages, biomarkers help identify and validate drug targets, ensuring that selected targets are biologically relevant and therapeutically actionable.
Preclinical Development
In preclinical models, biomarkers are used to:
- Assess pharmacological activity
- Evaluate dose-response relationships
- Monitor toxicity and safety signals
- Confirm mechanism of action
Clinical Trial Optimization
In clinical development, biomarkers support:
- Patient stratification (precision medicine approaches)
- Adaptive trial designs
- Early efficacy prediction
- Real-time treatment monitoring
Recent studies in Nature Reviews Drug Discovery emphasize that biomarker-guided trials significantly improve enrollment efficiency and increase the likelihood of demonstrating clinical benefit.
Surrogate Endpoints
In some cases, validated biomarkers can serve as surrogate endpoints, reducing trial duration and cost while maintaining scientific rigor.
Overall, biomarkers have evolved from supportive measurements to central components of clinical decision-making frameworks.
Challenges in Biomarker Discovery and Validation
Despite significant progress, biomarker development remains challenging, particularly in terms of reproducibility, validation, and translational reliability.
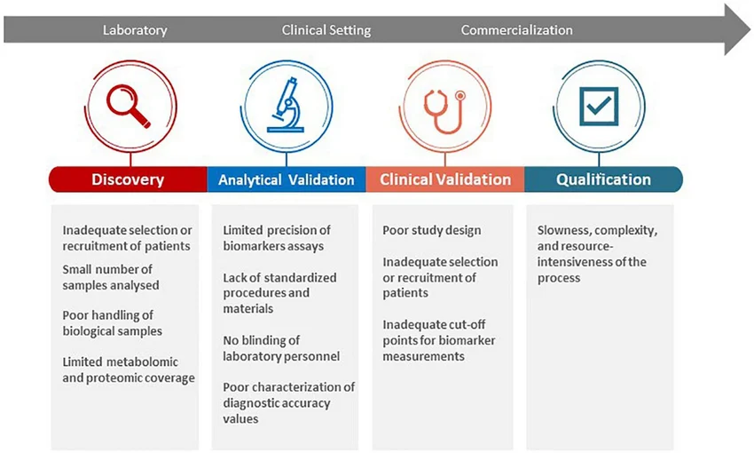
Biological Variability
There is immense biological variability between animal models and humans. Additionally, there is variability between patients and even tumors within the same patient.
Translational Gap
As stated in a recent article on translational medicine, "Despite tremendous investment…many biomarkers from basic science translate poorly to the clinic." There are many examples where animal models do not translate to humans due to biological variability. There can also be a lack of fidelity between the disease model and human disease.
Assay Standardization and Reproducibility
Recent reviews emphasize the need for robust assay validation. Variations in sample handling, detection methods, and analytical platforms can significantly impact biomarker reliability.
Regulatory Complexity
Regulatory agencies such as the FDA and EMA require clear context-of-use definitions and rigorous validation for biomarker qualification. This adds an additional layer of complexity to biomarker development programs.
Sample Quality Constraints
Many biomarker analyses require samples such as FFPE tissue, plasma, serum, etc. These can be heterogeneous and subject to degradation. Additionally, there can be limited samples available for analysis.
Stemming from these issues, it is crucial to have streamlined workflows when dealing with biomarkers. Choosing a CRO with experience in biomarker support is key.
Conclusion
Biomarkers are essential to modern drug discovery. They allow us to discover, create, and optimize therapeutics.
Not only are they used as biological indicators of a disease state but also as decision-making tools during drug development.
Through all stages of drug discovery, biomarkers make the process more targeted, efficient, and predictive. We can learn more about disease mechanisms and target validation in the early phase. All the way to monitoring clinical response and predicting efficacy in real time during clinical development. As technology like multi-omics and AI advance, we will continue to see an evolution in their use.
However, as you can see by some of the common biomarker challenges, there is still a need for tight experimental design and support. Contract research organizations have the resources and expertise to help you navigate through your biomarker needs.
Creative Bioarray Relevant Recommendations
| Products & Services | Description |
|---|---|
| PK/PD Biomarker Analysis | Creative Bioarray offers comprehensive PK/PD biomarker analysis to optimize dosing, predict efficacy, ensure safety, and accelerate drug development. |
References
- García-Gutiérrez MS, Navarrete F, et al. Biomarkers in Psychiatry: Concept, Definition, Types and Relevance to the Clinical Reality. Front Psychiatry. 2020. 11:432.
- Davis, K.D., Aghaeepour, N., et al. Discovery and validation of biomarkers to aid the development of safe and effective pain therapeutics: challenges and opportunities. Nat Rev Neurol. 2020. 16, 381-400.
- Landry V, Coburn P, et al. Diagnostic Accuracy of Liquid Biomarkers in Airway Diseases: Toward Point-of-Care Applications. Front Med (Lausanne). 2022. 9:855250.