Human Proximal Tubular Epithelial Cells
Cat.No.: CSC-C9228J
Species: Human
Source: Kidney
Cell Type: Epithelial Cell
- Specification
- Background
- Scientific Data
- Q & A
- Customer Review
Human proximal tubular epithelial cells (HPTECs) are primary cells isolated from the cortical region of the kidney, specifically lining the proximal convoluted tubule. As a critical component of the nephron, they are responsible for the majority of active solute reabsorption, including glucose, amino acids, and electrolytes, as well as the secretion of organic anions and cations. Moreover, HPTECs perform essential metabolic and endocrine functions, such as ammonia genesis, vitamin D hydroxylation, and erythropoietin synthesis.
A principal advantage of primary HPTECs over immortalized cell lines lies in their preservation of in vivo-like phenotypes. They retain characteristic polarity with well-developed apical brush borders, express high levels of drug transporters (e.g., OAT1, OAT3, OCT2, MATE1), and maintain key metabolic enzyme activities (e.g., γ-glutamyl transferase, alkaline phosphatase). Primary HPTECs also exhibit intact xenobiotic metabolism and stress response pathways, rendering them highly predictive for nephrotoxicity assessment.
From a translational perspective, HPTECs are widely employed in drug-induced kidney injury screening, transporter-mediated drug interaction studies, and renal disease modeling (e.g., acute kidney injury, diabetic nephropathy). Their human origin eliminates species-specific discrepancies commonly encountered with rodent models. Although they undergo limited proliferation in vitro, commercially available optimized culture systems support short-term expansion while retaining functional integrity. Thus, primary HPTECs represent the gold-standard cellular model for mechanistic nephrotoxicity and renal pharmacology research.
Human Proximal Tubular Epithelial Cell Interleukin-1 Receptor Signaling Triggers G2/M Arrest and Cellular Senescence During Hypoxic Kidney Injury
Hypoxia and interleukin (IL)-1β are independent mediators of tubulointerstitial fibrosis, the histological hallmark of chronic kidney disease (CKD). Here, we examine how hypoxia and IL-1β act in synergy to augment maladaptive proximal tubular epithelial cell (PTEC) repair in human CKD.
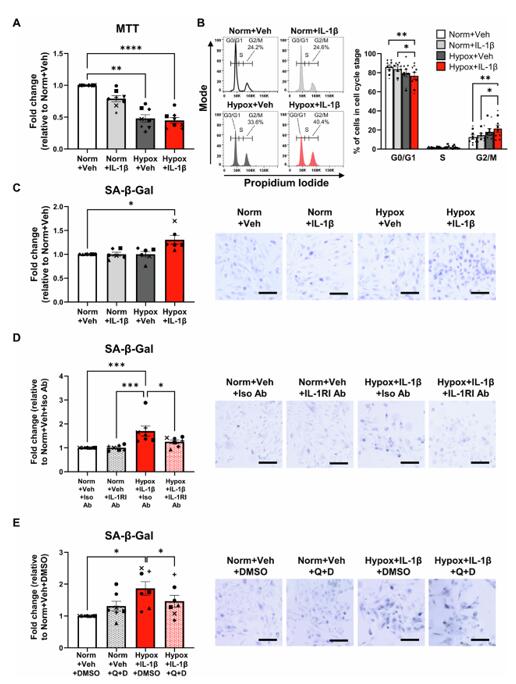
Ex vivo patient-derived PTECs were cultured under normoxic (21% O2) or hypoxic (1% O2) conditions in the absence or presence of IL-1β and examined for maladaptive repair signatures. Hypoxic PTECs incubated with IL-1β displayed a discrete transcriptomic profile distinct from PTECs cultured under hypoxia alone, IL-1β alone or under normoxia. Hypoxia+IL-1β-treated PTECs had 692 'unique' differentially expressed genes (DEGs) compared to normoxic PTECs, with 'cell cycle' the most significantly enriched KEGG pathway based on 'unique' down-regulated DEGs (including CCNA2, CCNB1 and CCNB2). Hypoxia+IL-1β-treated PTECs displayed signatures of cellular senescence, with reduced proliferation, G2/M cell cycle arrest, increased p21 expression, elevated senescence-associated β-galactosidase (SA-β-gal) activity and increased production of pro-inflammatory/fibrotic senescence-associated secretory phenotype (SASP) factors compared to normoxic conditions. Treatment of Hypoxia+IL-1β-treated PTECs with either a type I IL-1 receptor (IL-1RI) neutralizing antibody or a senolytic drug combination, quercetin+dasatinib, attenuated senescent cell burden.
These data identify a mechanism whereby hypoxia in combination with IL-1β/IL-1RI signaling trigger PTEC senescence, providing novel therapeutic and diagnostic checkpoints for restoring tubular regeneration in human CKD.
Ask a Question
Write your own review