Human Small Intestinal Smooth Muscle Cells
Cat.No.: CSC-C4860L
Species: Human
Source: Small Intestine; Intestine
Cell Type: Smooth Muscle Cell
- Specification
- Background
- Scientific Data
- Q & A
- Customer Review
Never can cryopreserved cells be kept at -20 °C.
Human Small Intestinal Smooth Muscle Cells (HSISMC) are primary cells isolated from human small intestine smooth muscle tissue. Smooth muscle cells are found in the circular and longitudinal muscle layers of the intestine and are responsible for controlling GI motility. Specifically, these muscle cells are responsible for peristalsis of the intestine, intestinal tone, and helping move luminal contents along. HSISMCSs often appear spindle shaped when grown in vitro and are known to express smooth muscle markers like α-SMA, desmin, and smooth muscle myosin heavy chain.
Physiologically, small intestine smooth muscle cells react to several neurotransmitters, growth hormones, and inflammatory cytokines. Smooth muscle cells are involved in signaling pathways that regulate contraction of the smooth muscle, cell proliferation, and extracellular matrix deposition. For this reason, these cells are often used to model disease processes that affect gut motility, intestinal fibrosis, and inflammation.
These cells being primary cells will have properties more closely related to smooth muscle in vivo when compared to an immortalized cell line. However, because these cells can only be passaged a limited number of times they are usually used at early passages. HSISMCs are a great cell type to use to study smooth muscle in vitro.
Rapid Automated Production of Tubular 3D Intestine-On-A-Chip with Diverse Cell Types using Coaxial Bioprinting
This study aimed to develop a novel coaxial bioprinting method to construct 3D tubular structures emulating small intestine organization and functionality. By analyzing bioink rheological properties and conducting biological validations, researchers sought to create a physiologically relevant model superior to conventional 2D systems for drug development applications.
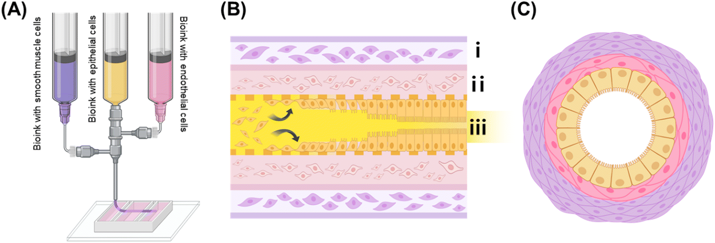
The intestine-on-a-chip (IOC) fabrication process is illustrated in Fig. 1, detailing the 3D extrusion-based coaxial bioprinting technique used to create tubular structures mimicking small intestine morphology. The exterior (purple) contains alginate-collagen bioink with human small intestine smooth muscle cells (HSISMC), the interior (pink) contains collagen bioink with EA.hy926, and the yellow portion represents gelatin bioink with Caco-2.
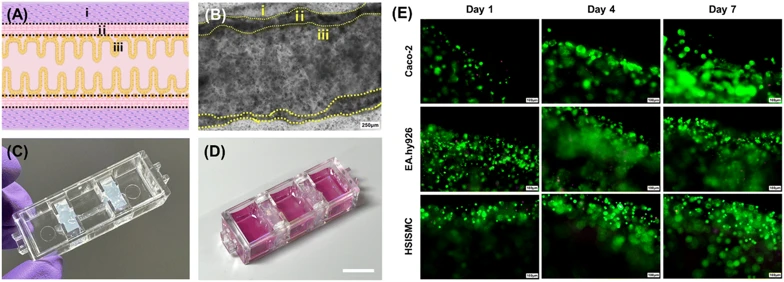
Post-printing, the gelatin inner layer liquefied, enabling cell mobility and adhesion to collagen-based walls via cell-binding functional groups. Fig. 2A shows the intended intestinal tubular structure. Microscopic imaging after 7-day co-culture confirmed successful adhesion of all three cell types (Fig. 2B). 3D confocal imaging immediately post-printing verified distinct epithelial, vascular, and muscle cell layering, establishing a perfusable 3D structure.
Fig. 2C illustrates the bioprinted tubular small intestine integrated onto the chip platform, forming the final IOC model. Calcein AM/ethidium homodimer staining of Caco-2 cells (Fig. 2D) demonstrated active epithelial proliferation with minimal cell death by day 7, indicating effective hollow channel formation without cytotoxicity. Endothelial and smooth muscle cells similarly showed robust growth in their designated layers (Fig. 2D). These results confirm successful co-culture replicating the physiological small intestine environment.
Ask a Question
Write your own review
- You May Also Need
