Rat Carotid Artery Smooth Muscle Cells
Cat.No.: CSC-C5020S
Species: Rat
Source: Artery
Cell Type: Smooth Muscle Cell
- Specification
- Background
- Scientific Data
- Q & A
- Customer Review
Rat Carotid Artery Smooth Muscle Cells (rCaASMCs) from Creative Bioarray are isolated from the rat carotid artery tissue. The method we use to isolate renal collecting duct epthelial cells was developed based on a combination of established and our proprietary methods. The rCaASMCs are characterized by immunofluorescence with antibodies specific to α-SMA. Each vial contains 0.5x10^6 cells per ml and is delivered frozen.
Rat Carotid Artery Smooth Muscle Cells (rCASMCs) are VSMCs obtained directly from the tunica media of the carotid artery. Typically, Sprague-Dawley or Wistar rats are used experimentally. RASMCs exhibit classic "hill-and-valley" morphology when confluent as well as a spindle shaped morphology with elongated nuclei when maintained in culture. RASMCs express α-SMA, SM-MHC, and calponin identifying them as VSMCs. They can phenotypically switch from this differentiated contractile phenotype to a proliferative synthetic phenotype that is migratory in response to certain disease stimuli. Phenotypic switching of this nature has been implicated in playing a large role in the pathology of these diseases. Rat is one of the most utilized organisms for cardiovascular research.
Traditionally, RASMCs have been used as a tool to study the cellular and molecular mechanisms of many vascular diseases. They have been used to study: Atherosclerosis (formation of foam cells, plaque stability), restenosis and neointimal hyperplasia following angioplasty, vascular remodeling in hypertension, and VSMC response to hemodynamic forces (shear stress, cyclic stretch). Screening for drugs that affect proliferation, migration, and contraction of VSMCs.
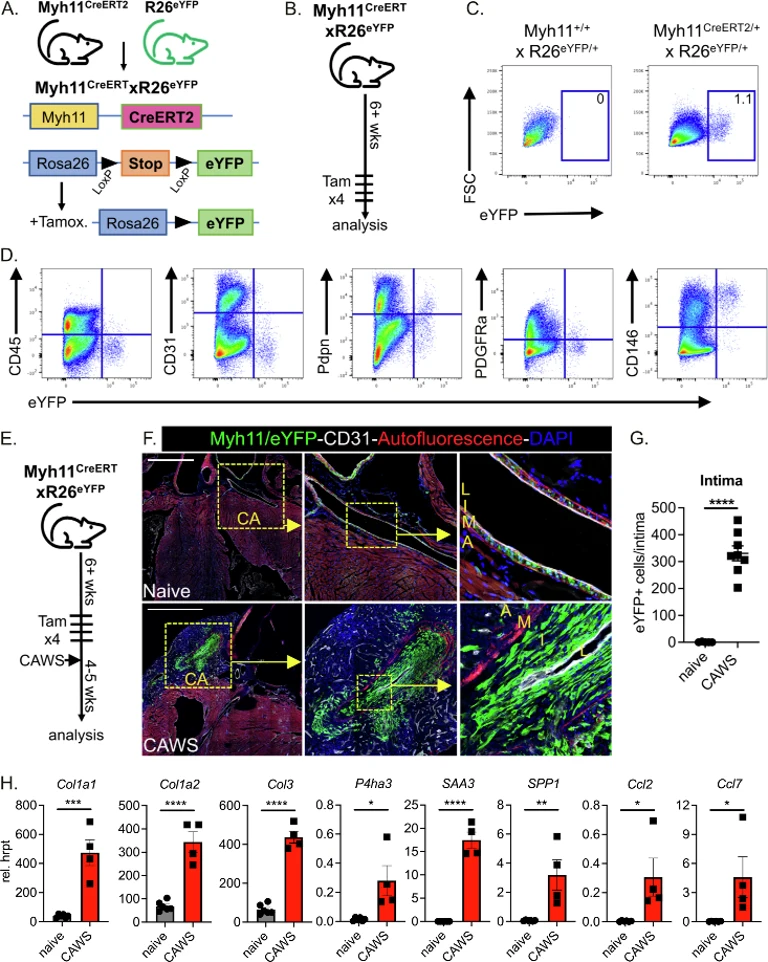
Luminal Myofibroblasts Develop from Myh11 + SMC Precursors
Luminal myofibroblast accumulation within inflamed vessels is a severe consequence of vasculitis that leads to arterial stenosis and ischaemia. Stock et al. explored the origin of these cells in Kawasaki disease (KD), a childhood coronary vasculitis. Using lineage tracing techniques in a murine KD model, they find that luminal myofibroblasts arise independently of adventitial fibroblasts and endothelial cells, and instead arise from smooth muscle cells (SMCs).
SMCs were traced using Myh11CreERT2 x R26eYFP mice, where tamoxifen-inducible labelling of Myh11+ SMCs was confirmed (Fig. 1A). Flow cytometry analysis confirmed that eYFP+ cells were present only in tamoxifen-treated mice (Fig. 1B, C). Phenotypic analysis showed the Myh11+/eYFP+ population was CD146+ and negative for endothelial (CD31), leukocyte (CD45), and fibroblast (Pdpn, PDGFRα) markers demonstrating specific labelling of SMCs (Fig. 1D).
To test if SMCs form luminal myofibroblasts during vasculitis, labelled mice were injected with CAWS and hearts analyzed 4-5 weeks later (Fig. 1E). In naive mice, Myh11+/eYFP+ cells remained confined to the medial layer (Fig. 2F). In contrast, CAWS-injected mice showed extensive Myh11-derived eYFP+ cells populating the thickened intima (Fig. 1F, G). Transcriptional analysis of sorted eYFP+ SMC-derived cells revealed upregulated fibrillar collagens (Col1a1, 1a2, Col3a1), collagen synthesis enzymes (P4HA3), inflammatory cytokines (SAA3, OPN), and chemokines (Ccl2/7) (Fig. 1H). These findings demonstrate that SMCs undergo media-to-intimal migration during vasculitis, acquiring fibrogenic and inflammatory properties that drive pathogenic luminal myofibroblast formation and vascular remodeling.
Ask a Question
Write your own review
- You May Also Need