Human Retinal Pericytes
Cat.No.: CSC-C4750Z
Species: Human
Source: Retina; Eye
Cell Type: Pericyte
- Specification
- Background
- Scientific Data
- Q & A
- Customer Review
Never can cryopreserved cells be kept at -20 °C
Human Retinal Pericytes (HRPs) are primary mural cells derived from human retinal microvasculature. Pericytes are crucial mediators of retinal vascular integrity and homeostasis. Pericytes intimately envelop endothelial cells within retinal capillaries where they facilitate blood-retinal barrier maintenance, capillary tone, and vascular maturation.
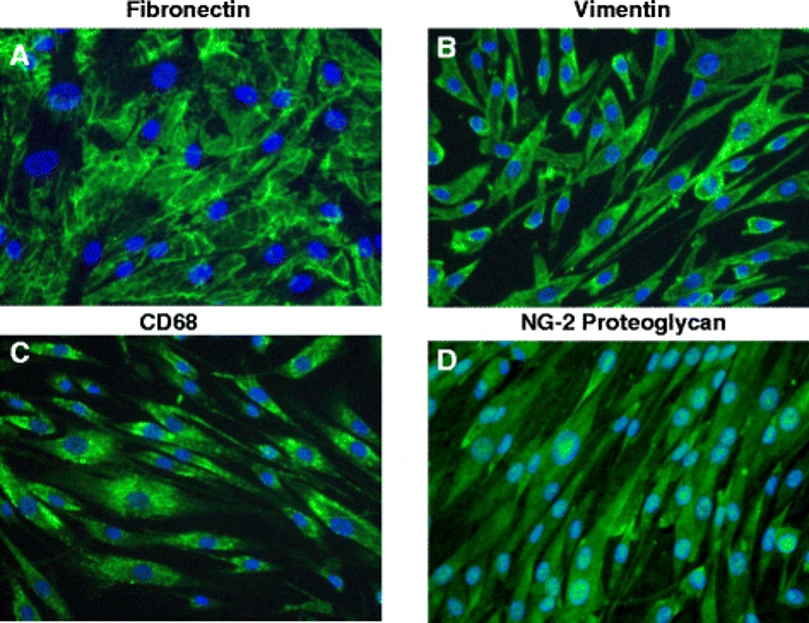
HRPs are spindle or stellate-shaped cells that culture as adherent monolayers under routine culture conditions. These cells display robust expression of pericyte markers such as platelet-derived growth factor receptor-β (PDGFR-β), NG2 (CSPG4), α-smooth muscle actin (α-SMA), and desmin. HRPs are also responsive to angiogenic and inflammatory stimuli including PDGF-BB, TGF-β, and VEGF and actively contribute to extracellular matrix deposition and vascular remodeling.
Global deletion of retinal pericytes recapitulates one of diabetic retinopathy's earliest observed clinical-pathological events described in classic histopathologic papers. As such, HRPs serve as a common model to study microvascular degeneration, endothelial-pericyte communication, and inflammation-induced vascular leakage associated with retinal disease. HRPs are also utilized in co-culture and 3D vascular models to study angiogenesis and barrier dynamics.
Small Extracellular Vesicles Derived from Human Retinal Pericytes under High Glucose and Hypoxia Conditions Promote Endothelial Cell Dysfunction In Vitro
Diabetic retinopathy (DR) is characterized by diabetic microvascular dysfunction within the retina. Poor communication between pericytes and endothelial cells (EC) plays a role in DR disease pathology. Nandeesh et al. researched small extracellular vesicles (sEV) released from diabetes-conditioned human retinal pericytes (HRP) characterized by high glucose [HG] + hypoxia and its effects on ECs.
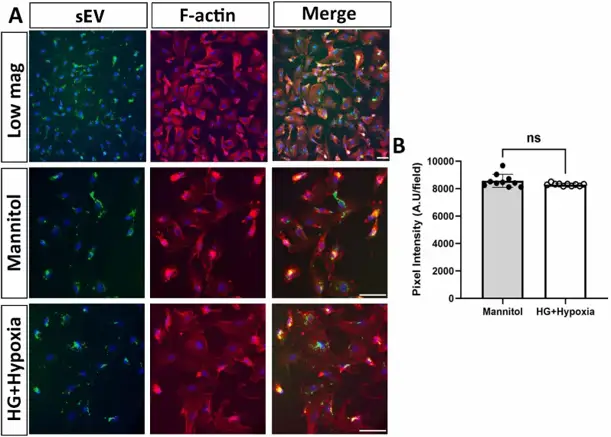
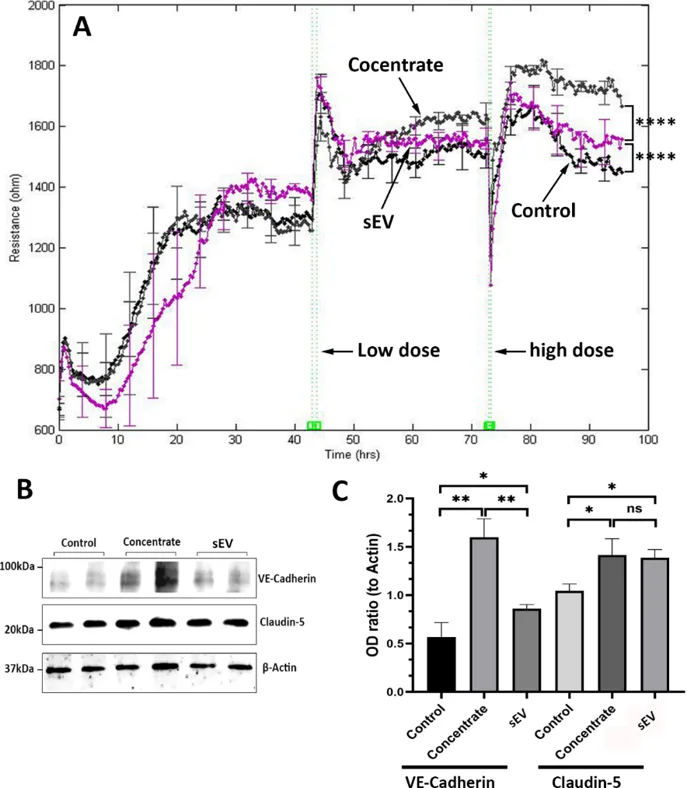
They first examined whether diabetes-like stress affects HRP sEV uptake by human retinal endothelial cells (HRECs). PKH67-labeled sEV (green) co-localized with DAPI-stained nuclei (blue) and phalloidin-stained F-actin (red), indicating efficient uptake (Fig. 1A). Quantification showed no significant difference in uptake efficiency between HG + hypoxia and control sEV (Fig. 1B). Retinal pericytes are crucial for blood-retinal barrier function. They hypothesized that pericyte-derived sEV contribute to HREC barrier integrity. Concentrated HRP-conditioned medium (CM) robustly increased HREC resistance measured by ECIS (Fig. 2A). Isolated sEV also enhanced resistance, though to a lesser extent than CM, and increased expression of barrier molecules VE-Cadherin and Claudin-5 (Fig. 2B, C), confirming sEV involvement in barrier function.
Ask a Question
Write your own review