Rat Cardiac Microvascular Endothelial Cells
Cat.No.: CSC-C2124
Species: Rat
Source: Heart
Cell Type: Endothelial Cell; Microvascular Cell
- Specification
- Background
- Scientific Data
- Q & A
- Customer Review
Rat Cardiac Microvascular Endothelial Cells (RCMECs) are primary endothelial cells harvested from the microvasculature of rat myocardium (Sprague-Dawley). RCMECs display normal endothelial cobblestone morphology, grow to confluency as a monolayer, and express endothelial markers such as CD31 (PECAM-1), von Willebrand Factor (vWF) and VE-cadherin. In addition, they have been shown to create capillary like tubular networks in vitro when plated on Matrigel. Because they are microvascular cells, they contain organ specific phenotypes that are not present in endothelial cells derived from larger vessels.
RCMECs are used to study cardiac-specific vascular functions and diseases. This includes uses such as: studying cardiac microcirculation, investigating angiogenesis and arteriogenesis during myocardial ischemia and infarction, modeling endothelial dysfunction during heart failure, determining cardiac endothelial cell response to inflammatory cytokines and hemodynamic forces and creating in vitro models of the cardiac microvascular barrier.
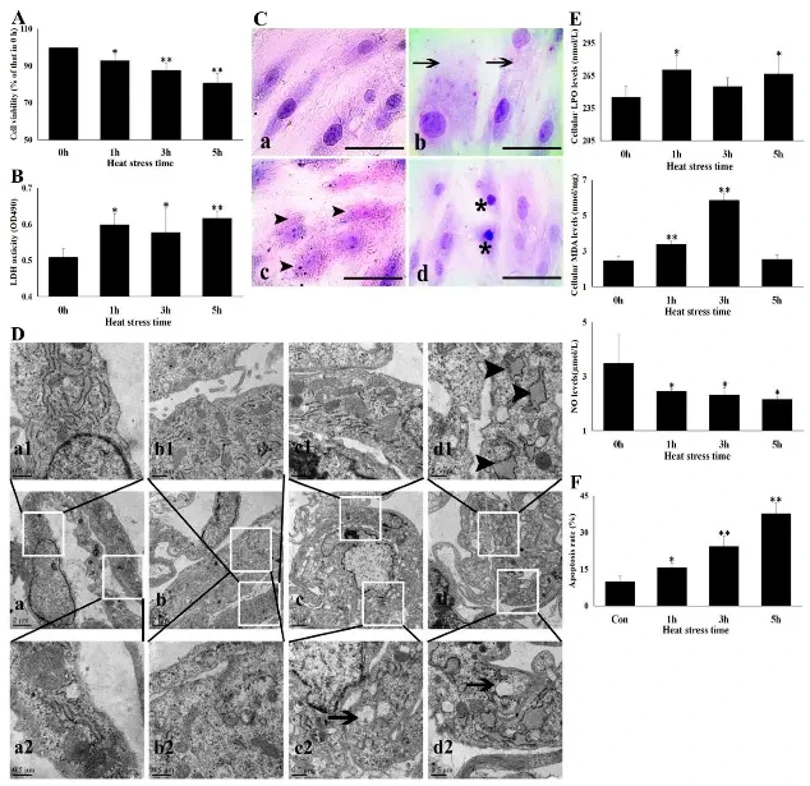
Heat Stress Damaged Cardiac Microvascular Endothelial Cells
Heat stress (HS) causes sudden death via heart failure, primarily through cardiac microvasculature contraction and myocardial ischemia. Cardiac microvascular endothelial cells (CMVECs) maintain vasodilatation, yet their molecular resistance mechanisms and aspirin's (ASA) protective role remain unclear. Zhang's team investigated HS-induced CMVEC injury and resistance mechanisms in vitro, and ASA's effects.
HS at 43°C significantly reduced CMVEC viability in a time-dependent manner (7.1% at 1 h, 19% at 5 h; Fig. 1A), with elevated LDH release indicating membrane damage (Fig. 1B). Light microscopy revealed progressive cellular swelling (1 h), granular degeneration and vacuolization (3 h), and necrosis with karyopyknosis (5 h; Fig. 1C). TEM showed mitochondrial cristae loss and endoplasmic reticulum swelling progressing to vacuolization (Fig. 1D).
HS induced oxidative stress, evidenced by increased intracellular LPO and MDA (Fig. 1E), and impaired endothelial function shown by reduced NO release from 1 h onward. Apoptosis increased sharply in a time-dependent manner (Fig. 1F). These results demonstrate that HS severely disrupts CMVEC homeostasis and function through oxidative damage, mitochondrial dysfunction, and apoptotic cell death.
Ask a Question
Write your own review