Non-human primate Bone Marrow CD34+ Cells
Cat.No.: CSC-C4445X
Species: Monkey
Source: Bone Marrow
Cell Type: CD34+ Cell
- Specification
- Background
- Scientific Data
- Q & A
- Customer Review
Non-Human Primate (NHP) Bone Marrow-derived CD34+ Hematopoietic Stem and Progenitor Cells (HSPCs) are the premier model for high-fidelity preclinical research. Given their profound genetic, immunologic, and physiological homology to humans, NHP CD34+ cells provide an unparalleled predictive framework for evaluating the safety and efficacy of next-generation therapeutics before clinical implementation.
- Unmatched Evolutionary Proximity: NHP CD34+ cells share critical cell surface markers, cytokine responses, and hematopoietic lineage hierarchies with human cells. This makes them indispensable for modeling hematopoietic reconstitution and immune system interactions that rodent models cannot accurately replicate.
- Gold Standard for Gene Editing & Viral Transduction: These cells are the benchmark for assessing the efficiency and long-term stability of CRISPR/Cas9, ZFN, and lentiviral delivery systems. They allow researchers to rigorously evaluate off-target effects and multi-lineage gene expression in a system that mirrors human marrow dynamics.
- Predictive Engraftment Kinetics: Our NHP CD34+ cells are validated for robust self-renewal and multilineage differentiation. They serve as a critical tool for autologous or allogeneic transplantation studies, providing precise data on neutrophil and platelet recovery times essential for protocol optimization.
- Rigorous Quality & High Purity: Isolated via advanced immunomagnetic separation, our cells achieve ≥90% purity and exceptional post-thaw viability. Each lot is sourced from healthy or diseased donors (Cynomolgus or Rhesus), ensuring consistent, reproducible performance for your most sensitive assays.
By utilizing our NHP CD34+ cells, you de-risk your R&D pipeline with a model that captures the complexity of human biology, accelerating the transition from bench to bedside.
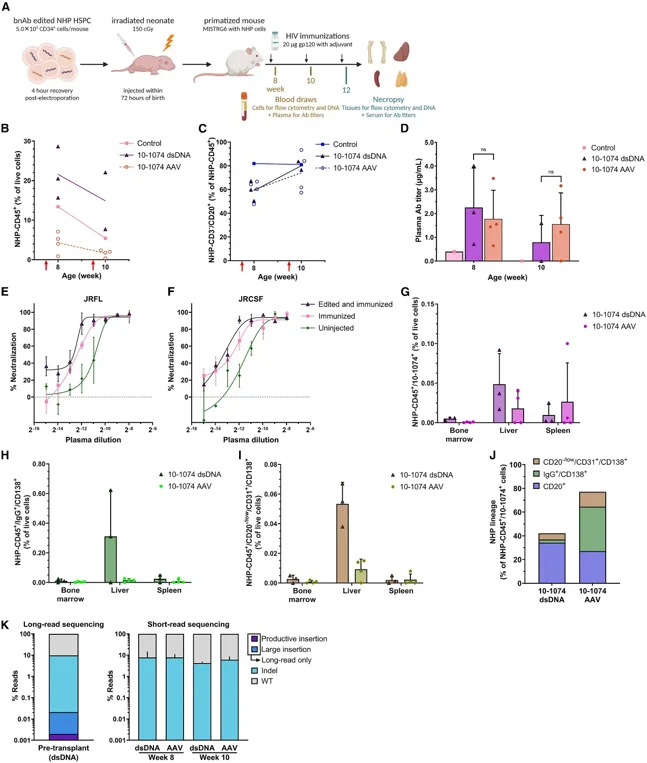
In Vivo Production of An Anti-HIV Antibody in Mice Engraftment of NHP HSPCs Following Non-Viral Knockin
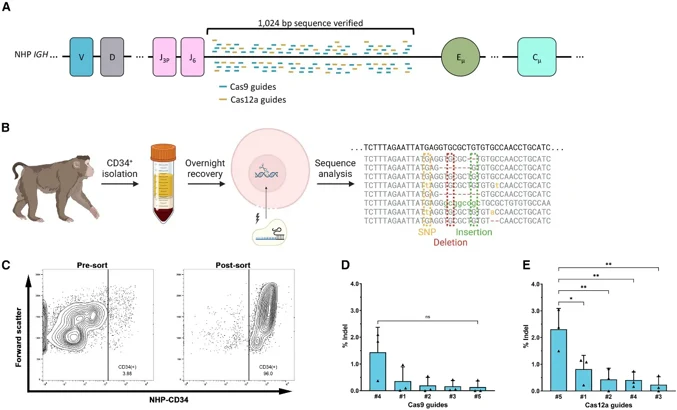
Gene editing strategies that do not rely on viral vectors are being explored for their potential to support durable biologics production. While clinical trials have shown that adeno-associated virus encoding broadly neutralizing antibodies can protect against HIV, these interventions often yield limited, short-lived responses. The development of non-viral gene editing approaches in hematopoietic stem and progenitor cells holds promise for long-term antibody production.
In this study, we evaluated CRISPR-Cas9 and CRISPR-Cas12a for gene knockin at the immunoglobulin heavy chain locus in non-human primate (NHP) hematopoietic stem and progenitor cells (HSPCs). The results demonstrate feasibility of gene editing in NHP HSPCs, with Cas12a showing a slight advantage in editing efficiency over Cas9 at this locus.
Transplantation of edited NHP HSPCs into MISTRG mice led to engraftment, B cell differentiation, and transgene expression of a reporter transgene or anti-HIV antibody after gp120 antigen immunization with detectable titers in circulation. These findings demonstrate the feasibility of using non-viral knockin in HSPCs as a potential strategy for sustained biologics production in the treatment of chronic diseases. Future work will assess the efficacy of this approach in an NHP model of HIV infection.
Ask a Question
Write your own review