Human Synovial Membrane Fibroblasts
Cat.No.: CSC-C9339W
Species: Human
Source: Synovium
Morphology: Multipolar
Cell Type: Fibroblast; Synoviocyte
- Specification
- Background
- Scientific Data
- Q & A
- Customer Review
Human Synovial Membrane Fibroblasts (HSMF), also referred to as fibroblast-like synoviocytes (FLS) or Type B synoviocytes, are primary mesenchymal cells isolated directly from human synovial membrane tissue. As the principal structural and functional cells of the joint lining, HSMF play a central role in maintaining synovial homeostasis, producing hyaluronan and lubricin to ensure smooth articular motion, and orchestrating the extracellular matrix architecture of the synovium.
HSMF possess a well-defined and distinctive molecular identity, expressing a panel of validated synovial markers including cadherin-11, CD90 (Thy-1), CD55, VCAM-1, ICAM-1, vimentin, and type IV/V collagens, along with the enzymatic marker uridine diphosphoglucose dehydrogenase (UDPGD), which reflects their constitutive capacity for hyaluronan biosynthesis. This authentically human, tissue-specific phenotype makes HSMF significantly more physiologically relevant than immortalized or animal-derived alternative models.
A key advantage of HSMF is their functional versatility across diverse research paradigms. Under inflammatory stimulation, these cells recapitulate the aggressive synoviocyte phenotype characteristic of Rheumatoid Arthritis (RA) - upregulating pro-inflammatory cytokines (IL-1β, IL-6, TNF-α), matrix metalloproteinases (MMPs), and responding to Toll-like receptor (TLR) activation, thereby faithfully modeling RA-associated joint pathophysiology in vitro. This positions HSMF as the preferred cellular platform for arthritis disease modeling, anti-inflammatory drug screening, and joint destruction mechanism studies.
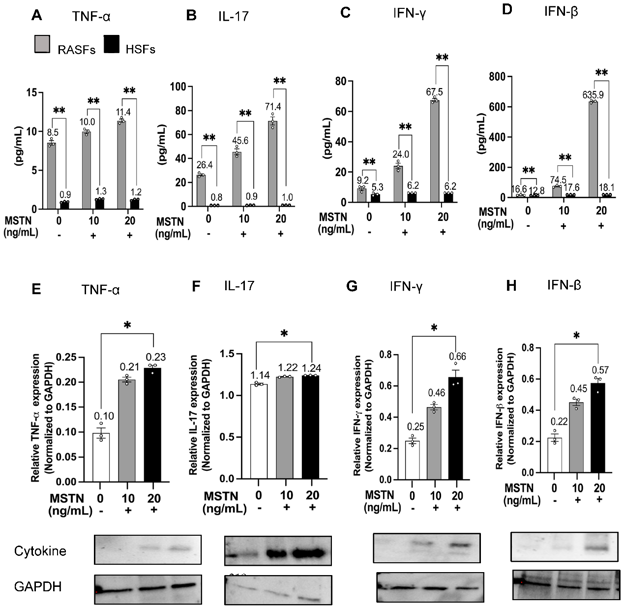
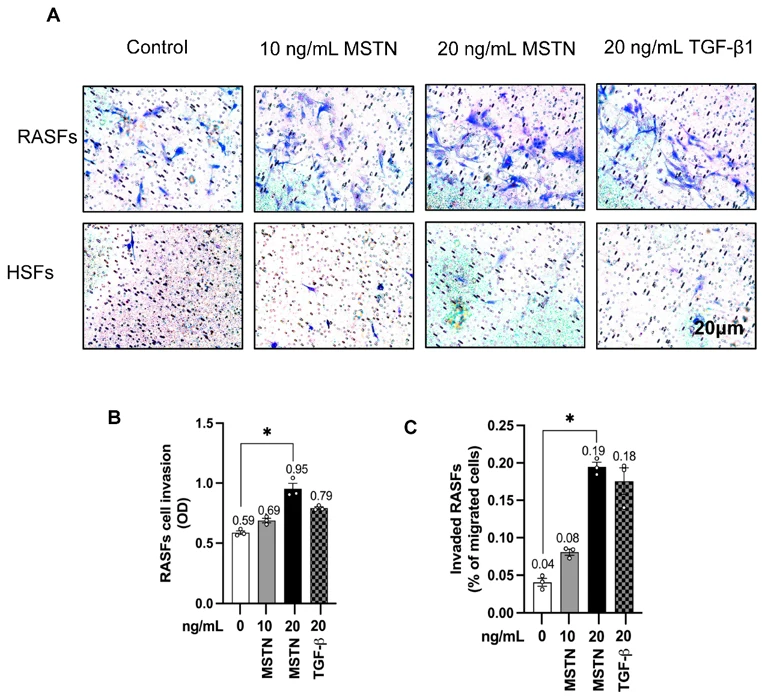
Myostatin Regulates Inflammatory Cytokine and Chemokine Expression, Rheumatoid Arthritis Synovial Fibroblast Invasion, and CD4+ Th Cell Transmigration
Rheumatoid arthritis synovial fibroblasts (RASFs) play a pivotal role in joint destruction in RA. Myostatin (MSTN), a myokine, is highly expressed in the RA synovium; however, its role in the function of RASFs is unclear. We hypothesized that MSTN amplifies inflammatory cytokines/chemokines, promotes RASF invasion, and facilitates CD4+ Th cell transmigration.
Immortalized MH7A cells (RASFs) and healthy synovial fibroblasts (HSFs) were treated with MSTN (0, 10, 20 ng/mL) for 0, 24, and 48 h. Cytokines (IL-8, IL-17, TNF-α, IL-6, IL-23, IFN-γ, IFN-β) and chemokines (CCL2, CCL20, CXCL13, CXCL1) were quantified by ELISA, RT-qPCR, and Western blotting. To evaluate MSTN regulation, cells were treated with pro-inflammatory mediators (TNF-α, IL-17, IFN-γ, IFN-β, CCL2, CXCL1). MSTN's effects on Thy-1(CD90)+ RASF/HSF proliferation, RASF invasion, and CD4+ T-cell transmigration were assessed.
Compared with HSFs, RASFs exhibited greater proliferative activity. MSTN significantly upregulated cytokines/chemokines, with CXCL1 showing the strongest induction in RASFs. IFN-γ and IL-17 robustly increased MSTN expression, indicating a feed-forward loop. MSTN did not alter Thy-1(CD90)+ fibroblast proliferation but significantly enhanced RASF invasion and CD4+ T-cell transmigration. Neutralizing CXCL1 or IL-17 reduced transmigration, with stronger inhibition via CXCL1. These findings offer new insights into the role of MSTN in RA pathogenesis and highlight its potential as a therapeutic target.
Different brands of dish or flask have different coating polymer and different manufacturing procedures, which do not affect most of the cells, but a small number of cells may have significant growth differences due to the use of different brands of dish or flask.
Ask a Question
Average Rating: 5.0 | 1 Scientist has reviewed this product
Consistent experimental results
The experimental results with IMR-5 are consistent and the product is available at a good price.
23 Jan 2022
Ease of use
After sales services
Value for money
Write your own review