Human Synovial Fluid Membrane Fibroblasts-RA
Cat.No.: CSC-C5436S
Species: Human
Source: Synovium
Cell Type: Fibroblast; Synoviocyte
- Specification
- Background
- Scientific Data
- Q & A
- Customer Review
Never can cryopreserved cells be kept at -20 °C
Human Synovial Membrane Fibroblasts derived from Rheumatoid Arthritis (RA-FLS, also termed Rheumatoid Arthritis Fibroblast-Like Synoviocytes, RASF) represent the most physiologically authentic and disease-relevant in vitro model for studying the pathogenesis and therapeutic intervention of rheumatoid arthritis. Isolated directly from the synovial tissue of confirmed RA patients, these primary human cells carry a stably "imprinted" aggressive phenotype that faithfully recapitulates the hallmark molecular and functional characteristics of the inflamed RA joint - a critical distinction from normal synovial fibroblasts or heterologous cell line surrogates.
A defining advantage of RA-FLS lies in their constitutively activated pro-inflammatory profile. Without requiring exogenous stimulation, these cells spontaneously secrete elevated levels of key pathological mediators including IL-6, MCP-1/CCL2, GM-CSF, and CXCL8, with MCP-1 production reported to be up to 16-fold higher than normal fibroblasts. They further express RANKL (driving osteoclastogenesis and bone erosion) and Dickkopf-1 (suppressing osteoblast-mediated bone repair), accurately mirroring the dual mechanism of joint destruction observed clinically in RA patients.
RA-FLS also possess a characteristically invasive and pannus-forming capacity, producing abundant matrix metalloproteinases (MMPs) that digest cartilage extracellular matrix, escaping contact inhibition in a tumor-like manner. Their surface immunophenotype - predominantly PDPN+THY1+/CD45-/CD34- - enables clean characterization and experimental stratification. Multiple dysregulated intracellular signaling axes, including IL-6/JAK-STAT, NF-κB, p38 MAPK, TLR, and Notch pathways, remain active and exploitable in culture, making RA-FLS an unparalleled platform for mechanistic dissection and drug target validation.
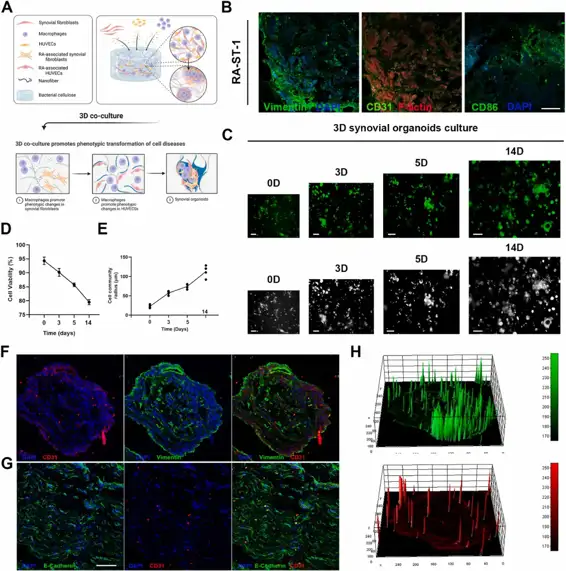
Constructing A 3D Co-Culture In Vitro Synovial Tissue Model for Rheumatoid Arthritis Research
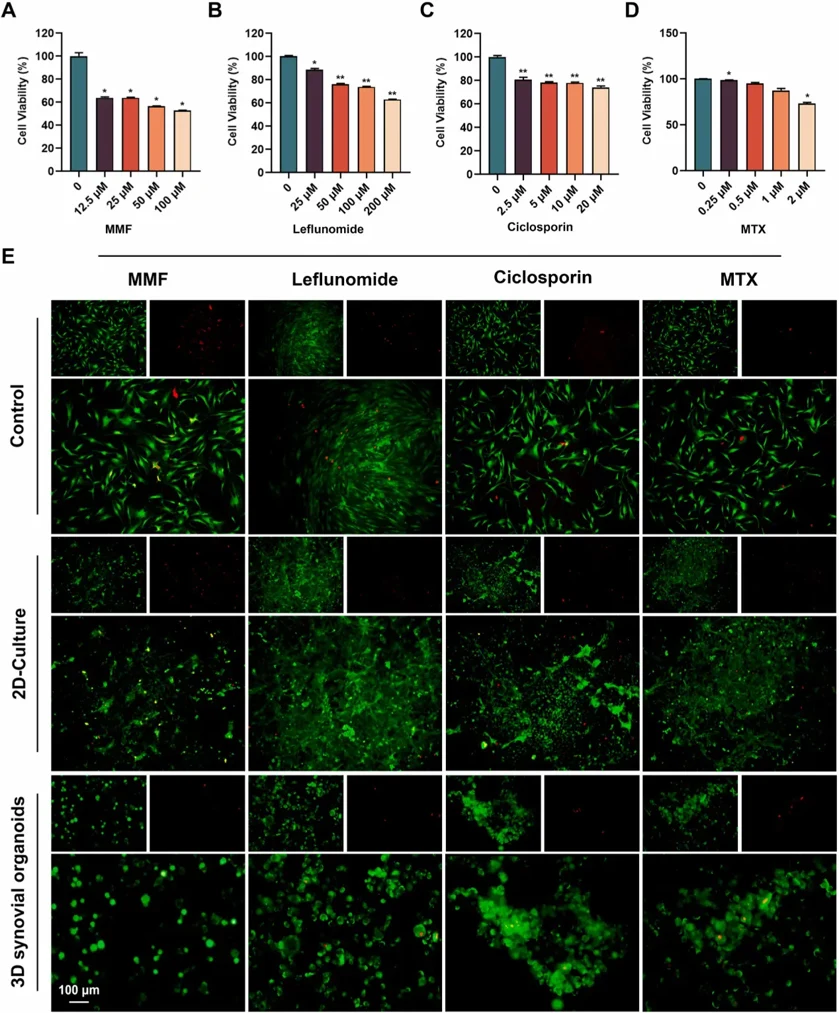
The development and exploration of highly effective drugs for rheumatoid arthritis remains an urgent necessity. However, current disease research models are no longer sufficient to meet the rapid development of high-throughput drug screening. In this study, bacterial cellulose simulating the structure of extracellular matrix was used as a 3D culture platform, and THP-1-derived M1 macrophages, representing the inflammatory component, human umbilical vein endothelial cells (HUVECs), simulating the vascular component, and rheumatoid arthritis fibroblast-like synoviocytes (RA-FLSs), embodying the synovial pathology, were co-cultured to simulate the pathological microenvironment in RA synovial tissues, and synovial organoids were constructed. Under three-dimensional (3D) culture conditions, there was a notable upregulation of fatty acid-binding protein 4 (FABP4) in polarized macrophages, and an enhancement of pathological phenotypes in HUVECs and RA-FLSs, mediated through the PI3K/AKT signaling pathway, including cell proliferation, migration, invasion and vascularization. Compared to planar cultures and 2D co-cultures, 3D synovial organoids not only exhibit a broader range of transcriptomic features characteristic of rheumatoid arthritis but also demonstrate increased drug resistance, likely due to the more complex and physiologically relevant cell-cell and cell-matrix interactions present in 3D environments. This model offers a promising path for personalized treatment, accelerating precision medicine in rheumatology.
Ask a Question
Write your own review