Hamster (Golden Syrian) Hepatocytes, Suspension
Cat.No.: CSC-C9357W
Species: Hamster
Source: Liver
Morphology: Spheric
Cell Type: Hepatocyte
- Specification
- Background
- Scientific Data
- Q & A
- Customer Review
Hamster (Golden Syrian) Hepatocytes, Suspension are freshly isolated primary liver parenchymal cells derived from Golden Syrian hamsters (Mesocricetus auratus), provided as a single-cell suspension. They maintain many of the morphological and functional properties of differentiated hepatocytes and serve as a physiologically relevant in vitro system for studying liver metabolism and toxicity.
Hamster hepatocytes cultured in suspension have a rounded shape. Following isolation, the cells are highly viable and express hepatocyte markers. They possess functional metabolic enzyme systems, including cytochrome P450 (CYP) enzymes, phase II conjugation enzymes, and uptake and efflux transporters. Consequently, they are useful for evaluating species-specific drug metabolism, metabolic stability, and metabolic profiles.
Hamster hepatocytes have been employed in various studies, including hepatotoxicity, drug-drug interactions, enzyme induction/inhibition, viral hepatitis, and lipid and glucose metabolism related to metabolic diseases. Because they are from a nonrodent species, metabolically competent, and readily cultured in suspension, Hamster (Golden Syrian) Hepatocytes allow for easy extrapolation to in vivo hamster studies.
The Feasibility of Establishing a Hamster Model for HBV Infection: In Vitro Evidence
Chronic HBV infection remains incurable, with research hampered by lack of immunocompetent small animal models. Here, Zhang's team explored the golden Syrian hamster as a potential model, beginning with in vitro assessment of HBV replication in primary hamster hepatocytes (PHaHs) via adenoviral HBV (Ad-HBV) transduction.
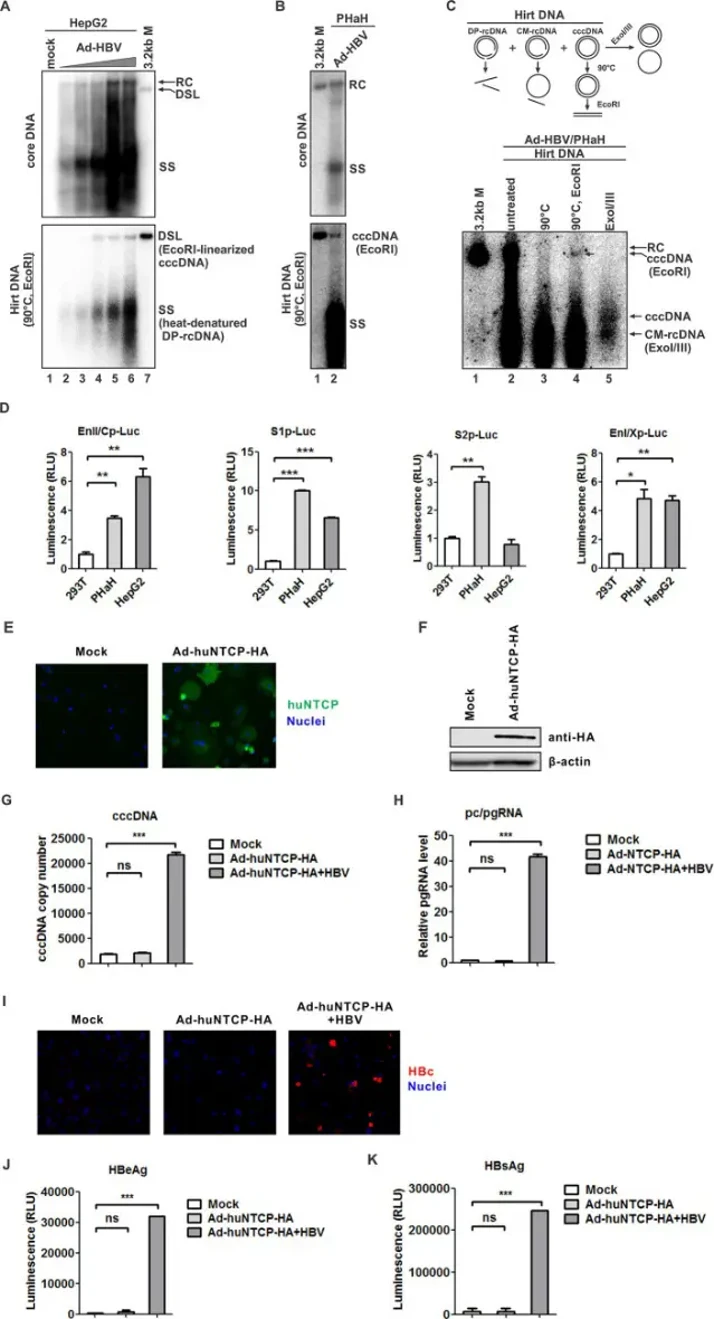
Due to low PHaH transfection efficiency, they employed adenoviral delivery. In HepG2 controls, Ad-HBV dose-dependently induced HBV DNA replication and cccDNA formation (Fig. 1A). Remarkably, cccDNA was readily detected in PHaHs (Fig. 1B), appearing as a 3.2 kb band after heat treatment and EcoR I linearization (Fig. 1C, lanes 1-4). Authenticity was confirmed by ExoI/III exonuclease resistance, leaving undigested cccDNA and CM-rDNA (Fig. 1C, lane 5)-both methods effectively distinguishing cccDNA from other HBV DNA species. These results demonstrate that PHaHs support HBV cccDNA formation via intracellular recycling. They next assessed HBV promoter activity by luciferase assay, with HepG2 and 293T cells as positive and negative controls. PHaHs supported all four major HBV enhancers/promoters (EnII/Cp, S1p, S2p, EnI/Xp) at levels comparable to HepG2 (Fig. 1D). Notably, S2p showed non-liver-specific activity as previously reported, but was somewhat more active in PHaHs. These data indicate PHaHs possess essential hepatic transcription factors for HBV transcription.
Ask a Question
Write your own review