Cryopreserved Plateable RAT Hepatocytes, min of 4 million cells/vial
Cat.No.: CSC-7662W
Species: Rat
Source: Liver
Cell Type: Hepatocyte
- Specification
- Background
- Scientific Data
- Q & A
- Customer Review
Cryopreserved Plateable Rat Hepatocytes represent the definitive in vitro model for evaluating hepatic metabolism, drug-drug interactions (DDI), and long-term cytotoxicity. Unlike suspension cells, our plateable hepatocytes are specifically optimized to re-establish a confluent, polarized monolayer in culture, mimicking the complex microenvironment of the liver parenchyma with high physiological fidelity.
- Long-Term Metabolic Stability: These hepatocytes maintain high levels of Phase I (CYP450) and Phase II (UGT, SULT) enzyme activities for up to 5-7 days in culture. This extended window is critical for identifying low-clearance compounds and metabolites that shorter assays might miss.
- Biliary Excretion & Transporter Activity: Our proprietary cryopreservation and recovery protocols ensure the preservation of key uptake (OATP, NTCP) and efflux (MDR1, MRP2) transporters. This makes them an ideal platform for studying hepatobiliary transport and cholestatic potential.
- Superior Attachment & Morphology: Validated for high attachment efficiency (>80%), these cells form robust bile canaliculi-like structures and maintain clear cell-cell boundaries, ensuring reproducible data across multiple plates and lots.
- Species-Specific Extrapolation: Utilizing high-quality Sprague-Dawley or Wistar strains, these cells provide the essential benchmark for in vitro to in vivo extrapolation (IVIVE), allowing for direct comparison with preclinical rat safety data.
By utilizing our cryopreserved plateable hepatocytes, you eliminate the variability and scheduling constraints of fresh tissue isolation while gaining a standardized, ready-to-use system for high-confidence toxicity screening.
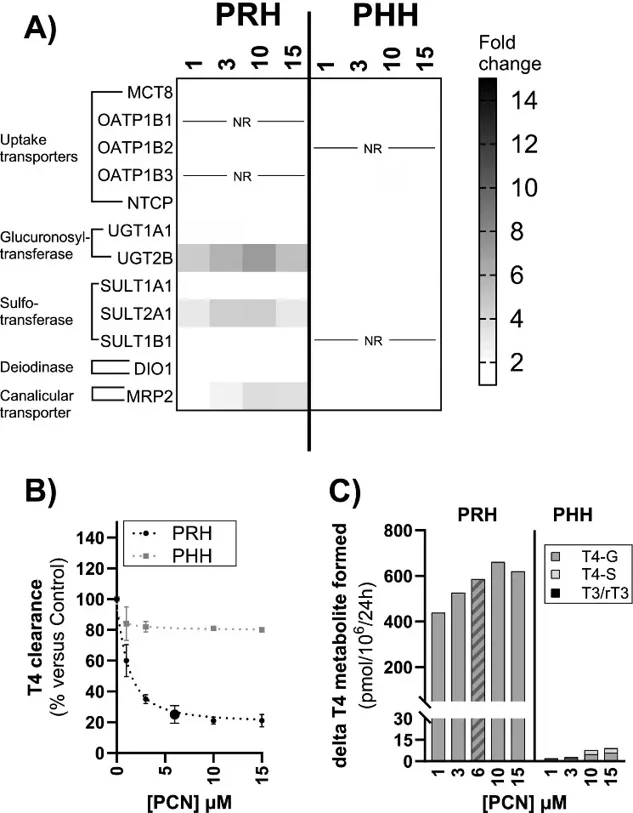
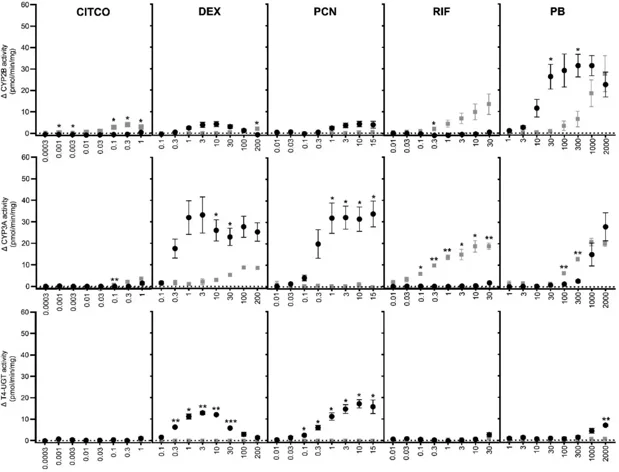
Differences in Thyroxine (T4) Metabolism Following In Vitro Exposure of Wistar Rat and Human Hepatocytes to CAR/PXR Activators
The species differences in thyroxine (T4) metabolism regulation by CAR/PXR activators were investigated using cryopreserved primary Wistar rat hepatocytes (PRH) and human hepatocytes (PHH) in 2D-sandwich over a 7-day treatment period.
Daily exposure of PRH to phenobarbital, 5-Pregnen-3β-ol-20-one-16α‑carbonitrile (PCN) or dexamethasone increased T4 clearance over the last 24 h exposure (up to 60 %, 79 % and 67 % over control, respectively) and secretion of T4-glucuronide (T4-G; up to 463, 661 and 545 pmol/106 cells over control, respectively). Effects were concentration-dependent for phenobarbital and PCN and highest at the lowest concentration for dexamethasone, while rifampicin barely affected T4 clearance and T4-G secretion. None of the compounds, at any tested concentration, affected these parameters in PHH. Additionally, mRNA expression data were consistent with the species-specific and concentration-dependent regulation of phase I Cyp/CYP, phase II Ugt/UGT and phase III Mrp2/MRP2 pathways occurring in rat and human liver following CAR/PXR activation. T4-UGT relative activity increased in PRH only, specifically by PCN, dexamethasone and phenobarbital. The comparison of PRH and PHH responses to compounds represents an important step towards using in vitro methods to reduce animal testing.
Ask a Question
Write your own review