Rabbit Small Intestine Crypt Epithelial Cells
Cat.No.: CSC-C5270S
Species: Rabbit
Source: Small Intestine; Intestine
Cell Type: Epithelial Cell
- Specification
- Background
- Scientific Data
- Q & A
- Customer Review
Rabbit small intestinal crypt epithelial cells from Creative Bioarray are isolated from the rabbit small intestinal tissue.The method we use to isolate rabbit small intestinal crypt epithelial cells was developed based on a combination of established and our proprietary methods. The rabbit small intestinal crypt epithelial cells are characterized by immunofluorescence with antibodies specific to pan-cytokeratin (PCK). Each vial contains 0.5x10^6 cells per ml and is delivered frozen.
Rabbit Small Intestine Crypt Epithelial Cells (RCI) are primary epithelial cells derived directly from intestinal crypts of Lieberkühn and are undifferentiated with high proliferative capacity and stem/progenitor cell properties. The cells continuously renew the intestinal villi, differentiating into all epithelial lineages (enterocytes, goblet, enteroendocrine, Paneth cells etc.). On Matrigel these cells self-organize into three-dimensional structures called organoids that model the crypt-villus axis as well as cellular hierarchy. Rabbits are extensively used in GI physiology and disease modeling as well as for their role in nutrient absorption.
As such their broadest application is in GI biology and disease modeling. Examples include studies of intestinal stem cells and regulation of epithelial differentiation, intestinal host-pathogen interactions (with enteric viruses, E. coli, etc.), intestinal mucosal immunology and nutrient transport. Additionally, the intestinal organoid model is being widely used for applications in toxicology (screening for effects on epithelial barrier function) and regenerative medicine. Being a primary cell line, cells will vary between donors in terms of viability, ability to grow in culture and differentiate. Specialized media can maintain stemness of these cells.
L-WRN CM Induces Less Proliferation and More Differentiation in Rabbit Caecum Organoids than Pharmacological Inhibitors
Intestinal organoids are self-organized 3D structures formed by polarized epithelial cells. While widely used in other species, this model had not been established in rabbits despite its potential for biomedical and veterinary research.
Mussard et al. established the first rabbit caecum organoid model from intestinal crypts using two niche reconstitution approaches: 2Ki medium (GSK3β/MEK inhibitors) promoted proliferation and stemness with high expression of Lgr5, Olfm4 and Pcna, whereas L-WRN CM induced differentiation, particularly at low concentration (50-5%), marked by elevated absorptive (Krt20, Vil1), secretory (Atoh1, Muc2, Pyy) and reduced stem cell markers.
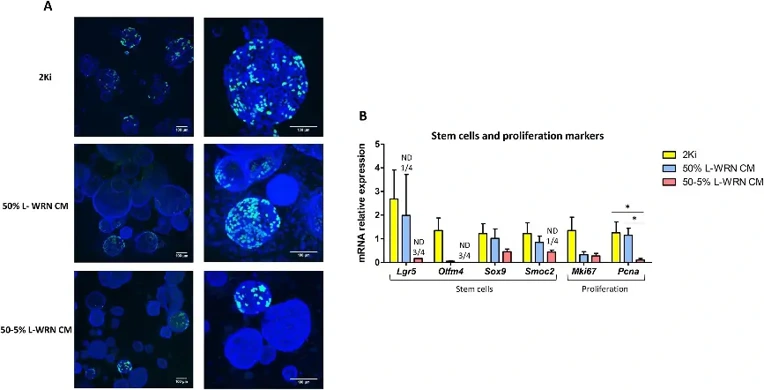
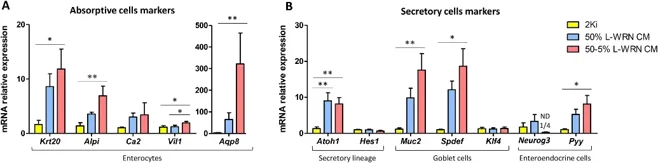
Non-spherical structures with thicker epithelium and smaller diameter in L-WRN CM cultures suggested reduced proliferation and enhanced differentiation (Merker et al., 2016). EdU staining confirmed fewer proliferating organoids in L-WRN CM conditions, with no EdU+ cells in non-spherical organoids (Fig. 1A). Stem cell markers (Lgr5, Olfm4, Sox9, Smoc2) showed highest expression in 2Ki medium, with Lgr5 undetectable in many L-WRN CM samples (Fig. 1B). The proliferation marker Pcna was significantly reduced in 50-5% L-WRN CM (Fig. 1B). Conversely, differentiation markers were elevated in L-WRN CM organoids. Absorptive cell markers (Krt20, Alpi, Vil1, Aqp8) and the secretory factor Atoh1 were significantly increased, as were goblet (Muc2, Spdef) and enteroendocrine (Pyy) markers, particularly at 50-5% concentration (Fig. 2A, B).
Ask a Question
Write your own review
- You May Also Need
