Mouse Retinal Ganglion Cells
Cat.No.: CSC-C5418S
Species: Mouse
Source: Retina; Eye
Cell Type: Retinal Ganglion Cell; Neuron
- Specification
- Background
- Scientific Data
- Q & A
- Customer Review
Mouse retinal ganglion cells from Creative Bioarray are isolated from the mouse retinal tissue. The method we use to isolate mouse retinal ganglion cells was developed based on a combination of established and our proprietary methods. The mouse retinal ganglion cells are characterized by immunofluorescence with antibodies specific to MAP-2. Each vial contains 0.5x10^6 cells per ml and is delivered frozen.
Mouse Retinal Ganglion Cells (mRGCs) serve as an indispensable in vitro model for studying axonal biology, neurodegeneration, and high-throughput drug screening. As the sole output neurons of the retina, RGCs are uniquely positioned at the intersection of ophthalmology and central nervous system (CNS) research, making them the premier choice for modeling diseases such as glaucoma, optic neuropathies, and traumatic brain injury.
- Physiological Relevance & Genetic Consistency: Sourced from validated murine models, our mRGCs maintain the complex morphological and electrophysiological characteristics of primary CNS neurons, offering superior biological translation compared to immortalized cell lines.
- High Purity & Viability: Utilizing advanced immunopanning or magnetic-activated cell sorting (MACS) targeting Thy1.2 or molecular markers like RBPMS, our cells reach a purity of >95%. This ensures that experimental data is representative of neuronal response without interference from glial or fibroblastic contamination.
- Robust Axonal Outgrowth: Our optimized primary culture protocols yield mRGCs with exceptional regenerative capacity. They exhibit rapid neurite extension and synaptogenesis within 48-72 hours, providing an ideal platform for evaluating neuroregenerative compounds and cytoskeletal dynamics.
- Ready-to-Use Versatility: Validated for various downstream applications, including calcium imaging, patch-clamping, and spatial transcriptomics, these cells streamline your R&D pipeline from target discovery to preclinical validation.
By integrating our high-performance mRGCs into your workflow, you gain a reliable, scalable, and highly reproducible system to accelerate your breakthroughs in neuroprotection and vision restoration.
Exogenous Hydrogen Sulfide and NOX2 Inhibition Mitigate Ferroptosis in Pressure-Induced Retinal Ganglion Cell Damage
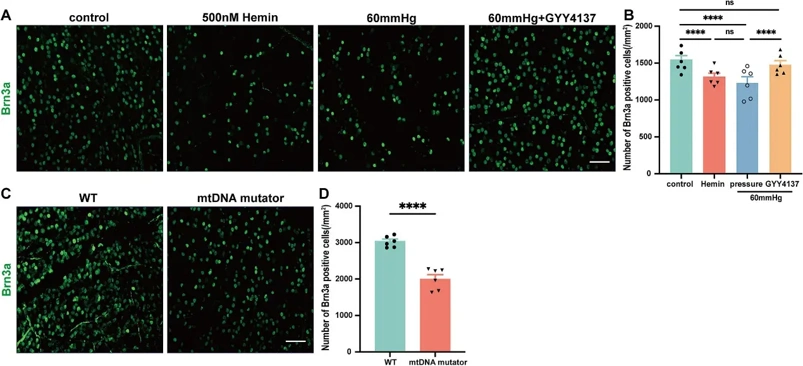
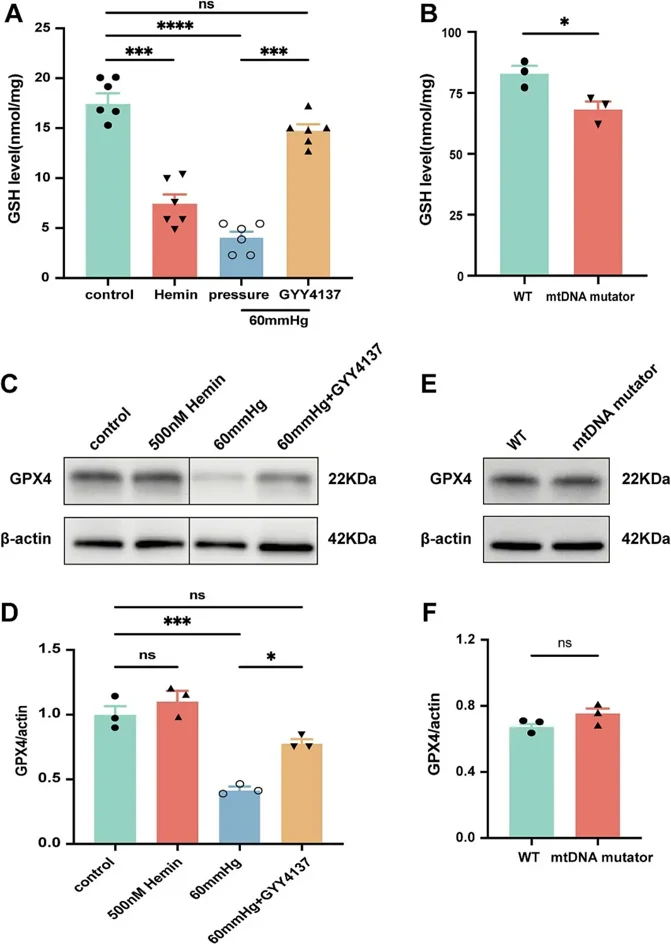
Glaucoma, a leading cause of irreversible blindness worldwide, is characterized by the progressive degeneration of retinal ganglion cells (RGCs). The mechanisms underlying RGCs loss in glaucoma remain unclear, but ferroptosis-an iron-dependent form of oxidative cell death-has been implicated, particularly in intraocular pressure (IOP)-induced RGCs loss. This study aims to investigate the interplay between elevated pressure, mitochondrial dysfunction, iron homeostasis, and ferroptosis in RGCs death, focusing on how hydrogen sulfide (H2S) may mitigate pressure-induced ferroptosis and protect RGCs.
In vitro, we confirm that elevated pressure, iron overload, and mitochondrial dysfunction lead to RGCs loss, increased retinal ferrous iron and total iron content, and heightened reactive oxygen species (ROS). Notably, pressure increases NADPH oxidase 2 (NOX2) and decreases glutathione peroxidase 4 (GPX4), a key regulator of ferroptosis. NOX2 deletion or inhibition by H2S prevents pressure-induced RGCs loss and ferroptosis. Our findings reveal that H2S chelates iron, regulates iron metabolism, reduces oxidative stress, and mitigates ferroptosis, positioning slow-releasing H2S donors are positioning as a promising multi-target therapy for glaucoma, with NOX2 emerging as a key regulator of ferroptosis.
Ask a Question
Write your own review