Human Prostate Fibroblasts (HPrF)
Cat.No.: CSC-7733W
Species: Human
Source: Prostate
Cell Type: Fibroblast
- Specification
- Background
- Scientific Data
- Q & A
- Customer Review
Human Prostate Fibroblasts (HPrF) are a primary culture of fibroblasts derived from human prostate tissue. HPrFs can be used as an in vitro model system relevant to prostate stromal biology and epithelial-stromal interactions. Fibroblasts within the prostate microenvironment are critical mediators of extracellular matrix production, tissue remodeling, and paracrine regulation of prostate epithelial cells.
HPrF cells are spindle-shaped and appear fibroblast-like when observed microscopically. HPrFs can form adherent monolayers when maintained in culture. HPrF cells express fibroblast markers and actively secrete extracellular matrix components including collagen and fibronectin. Like other fibroblast populations, HPrF cells respond to various stimuli such as androgen stimulation and cytokine-mediated signaling pathways. These pathways have both normal roles in prostate growth and development as well as pathological roles in disease.
Human Prostate Fibroblasts have been used as a tool to study the role of stromal cells in many prostate diseases including benign prostatic hyperplasia and prostate cancer. They have been used in co-culture with prostate epithelial cells to study fibroblast-epithelial cell crosstalk, regulation of the tumor microenvironment, and activation of cancer-associated fibroblasts. HPrFs have also been used for testing anti-fibrotic, anti-inflammatory and stroma-targeting therapeutics.
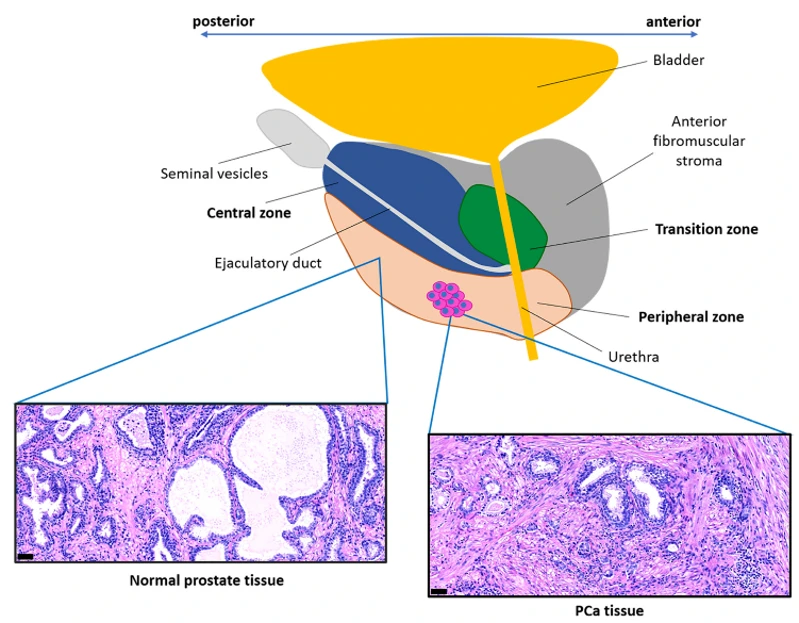
Identification of Functional and Diverse Circulating Cancer-Associated Fibroblasts in Metastatic Castration-Naïve Prostate Cancer Patients
Cancer-associated fibroblasts (CAFs) are major drivers of prostate cancer (PCa) progression and metastasis. However, whether they are present in circulation has not been investigated. Booijink et al. determined if there are circulating CAFs (cCAFs) present in the blood of metastatic castration-naïve PCa (mCNPC) patients and characterized phenotype and function.
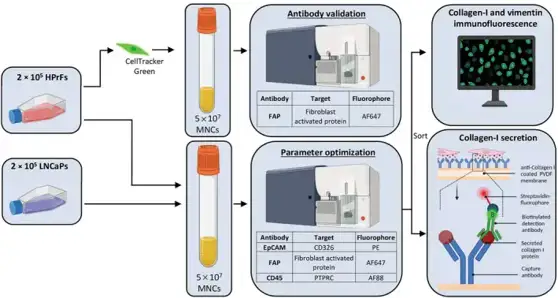
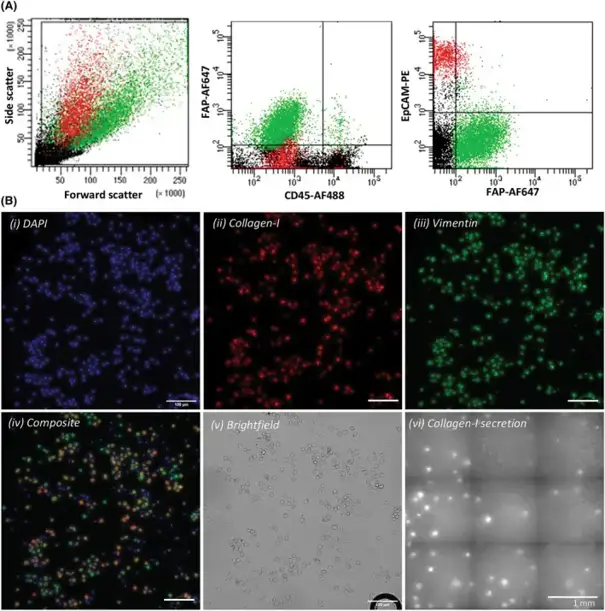
To isolate circulating cancer-associated fibroblasts (cCAFs), they developed a workflow combining immunofluorescence labeling, flow-activated cell sorting (FACS), and functional characterization (Fig. 1). Fibroblast-activated protein (FAP) was chosen as a CAF surface marker due to its identification on activated CAFs and association with prostate cancer (PCa) prognosis. Human primary prostate fibroblasts (HPrFs) and PCa cells were then spiked into human mononuclear cells (MNCs) to mirror patient samples. 2 × 105 HPrFs (cCAFs) and 2 × 105 LNCaP cells (circulating tumor cells, CTCs) were added to 5 × 107 MNCs. Samples were labeled with antibodies specific for FAP (cCAFs), CD45 (leukocytes), and EpCAM (CTCs). Cells were then sorted by FACS into FAP+ and FAP- fractions for downstream functional or phenotypic characterization, including intracellular staining for fibroblast markers vimentin and collagen-I and analysis of extracellular collagen-I secretion (Fig. 1). They were able to use FACS to successfully isolate FAP+EpCAM- HPrFs (Fig. 2A) and FAP- EpCAM+ LNCaPs. Immunofluorescence imaging showed sorted FAP+EpCAM- HPrFs were positive for collagen-I and vimentin expression. EpCAM+ LNCaPs were negative for both collagen-I and vimentin expression (Fig. 2B). Validation of function utilizing a collagen-I secretion assay showed numerous collagen-I spots present from sorted FAP+EpCAM- HPrFs whereas FAP- EpCAM+ LNCaPs revealed no spots, indicative of collagen-I secretion (Fig. 2B).
Ask a Question
Write your own review