C57BL/6 Mouse Oral Mucosal Epithelial Cells
Cat.No.: CSC-C9084J
Species: Mouse
Source: Oral Cavity
Cell Type: Epithelial Cell
- Specification
- Background
- Scientific Data
- Q & A
- Customer Review
C57BL/6 Mouse Oral Mucosal Epithelial Cells are primary epithelial cells isolated from the oral mucosa of pathogen-free C57BL/6 inbred mice. They serve as an important in vitro model for the study of oral mucosal biology and disease. In addition to displaying specific markers such as pan-cytokeratin, EpCAM, E-cadherin, and cytokeratin 5/14, the adherent cells have a distinctive polygonal, cobblestone-like epithelial morphology. Due to their slow development rate, these cells require a specialized epithelial medium, containing growth factors on surfaces coated with a matrix in order to be cultured in vitro for a maximum of two to four passes.
Studies on infectious disorders like candidiasis, inflammatory reactions, chemical-induced oral mucositis, and radiation-induced ulceration frequently use them as a physiologically appropriate model. Additionally, they are crucial in wound healing, assessing medication toxicity, and oral squamous cell carcinoma growth. Despite their transient nature and batch-to-batch variation, they are crucial in preclinical and translational oral research due to their pure genetic background and capacity to faithfully retain natural mucosal characteristics.
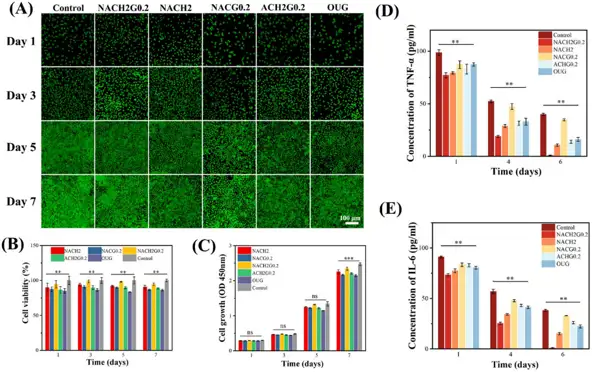
Biocompatibility and Anti-Inflammatory Assay of Bionic Microneedle Patch
Bacterial infectious stomatitis (BIS), characterized by severe ulcer infections, presents treatment problems due to protracted healing and patient pain. Qin et al. created a bioinspired hydrogel microneedle (MN) suction cup patch that combines wet adhesive qualities with precise drug delivery, inspired by the Drosophila tarsal claw.
Cytotoxicity analysis using the MTT assay and live/dead staining revealed that cell viability exceeded 85% for all hydrogel formulations, with the optimum NACH2G0.2 formulation exhibiting the best viability (Fig. 1B, C). Predominant green staining revealed great biocompatibility (Fig. 1A), demonstrating that these hydrogels maintain favorable antibacterial efficacy without causing detrimental physiological consequences. TNF-α and IL-6 levels were measured in mouse oral mucosal epithelial cells to assess their anti-inflammatory activity. By day 6, NACH2G0.2 reduced TNF-α and IL-6 to 0.90 and 0.94 pg mL-1, respectively, indicating near-complete inflammatory resolution. This was significantly superior to HACC or GSNO alone, and significantly lower than the commercial oral ulcer gel (OUG) group (16.05 and 22.31 pg mL-1) (Fig. 1D, E). These findings show that administering both HACC and GSNO has a higher therapeutic efficacy.
Ask a Question
Write your own review