Immortalized Human B Cells
Cat.No.: CSC-I9357Z
Species: Homo sapiens
Morphology: Polygonal
Culture Properties: Adherent
- Specification
- Background
- Scientific Data
- Q & A
- Customer Review
Note: Never can cells be kept at -20 °C.
Immortalized Human B Cells are laboratory-established cell lines derived from primary human B lymphocytes, modified to proliferate indefinitely while maintaining key B cell functions. Immortalization is commonly achieved through Epstein-Barr virus (EBV) transformation, which activates viral latent genes that promote survival and proliferation, or via genetic introduction of oncogenes such as c-MYC or BCL-2. This process allows the cells to bypass normal replicative senescence, providing a stable, renewable in vitro system for immunological research.
In culture, immortalized human B cells grow in suspension and often form clusters characteristic of lymphoid cells. They express canonical B cell markers, including CD19, CD20, CD21, and surface immunoglobulins, reflecting both their origin and functional identity. These cells retain the ability to produce antibodies, present antigens, and activate signaling pathways such as the B cell receptor (BCR) and NF-κB, making them a reliable model for studying B cell biology and immune responses.
Functionally, immortalized human B cells are widely used in immunology, infectious disease research, and therapeutic antibody development. They serve as models for B cell activation, differentiation, signaling, and viral infection, particularly EBV-related studies. They are also applied in vaccine research, monoclonal antibody discovery, and investigations of autoimmune disorders. Their long-term stability, reproducibility, and preservation of B cell phenotype make them a robust platform for both basic and translational immunology research.
Unbiased Screening of Immortalized B Cell Libraries Derived from Human PBMCs and Tonsils Produce B Cell Clones with Cross Reactive Neutralization to SARS-Cov-2
The continuous evolution of SARS-CoV-2 poses significant challenges to vaccine efficacy and therapeutic interventions, necessitating rapid, adaptable approaches to combat immune escape variants. Marsman et al. aimed to develop a robust platform using immortalized human B cell libraries from PBMCs and tonsil tissues to discover cross-reactive neutralizing antibodies.
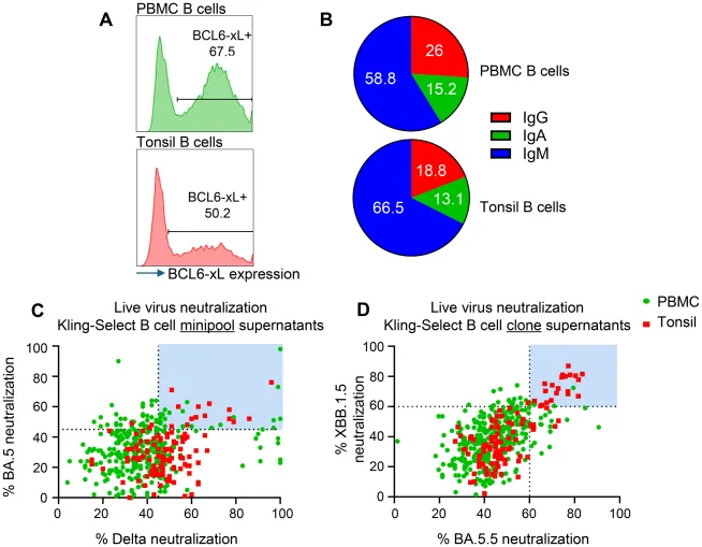
They generated immortalized B cell libraries from fresh human PBMCs and frozen tonsil tissue by retroviral transduction with BCL-6, BCL-xl, and GFP. Transduction efficiencies were 67.5% for PBMCs and 50.2% for tonsils (Fig. 1A), with the difference likely due to fresh versus frozen sample handling. Flow cytometry revealed diverse Ig isotypes in both libraries, with IgM predominating-particularly in tonsils, consistent with their role as naive B cell reservoirs. IgG comprised 26% (PBMC) and 19% (tonsils), while IgA represented 15% and 13%, respectively (Fig. 1B), indicating representative Ig repertoire capture.
'Minipools' (MPs) were generated by sorting 25 cells per well into ten 384-well plates, followed by 3-week expansion. MPs were screened for SARS-CoV-2 reactivity by binding to Spike and envelope proteins, yielding 474 positive MPs from PBMCs and 123 from tonsils. MPs with >45% neutralization against Delta and BA.5 variants (Fig. 1C) were single-cell sorted to establish monoclonal populations. From 440 monoclonal supernatants screened against XBB.1.5 and BA.5.5, most showed >50% neutralization, with 26 demonstrating >60% neutralization against both variants (blue box, Fig. 1D). Tonsil-derived B cells exhibited lower binding and hit rates but enriched cross-neutralizing clones (Fig. 1D). Sequencing of clones with >60% dual-variant neutralization identified 12 unique VH/VL pairs.
Ask a Question
Write your own review
- Adipose Tissue-Derived Stem Cells
- Human Neurons
- Mouse Probe
- Whole Chromosome Painting Probes
- Hepatic Cells
- Renal Cells
- In Vitro ADME Kits
- Tissue Microarray
- Tissue Blocks
- Tissue Sections
- FFPE Cell Pellet
- Probe
- Centromere Probes
- Telomere Probes
- Satellite Enumeration Probes
- Subtelomere Specific Probes
- Bacterial Probes
- ISH/FISH Probes
- Exosome Isolation Kit
- Human Adult Stem Cells
- Mouse Stem Cells
- iPSCs
- Mouse Embryonic Stem Cells
- iPSC Differentiation Kits
- Mesenchymal Stem Cells
- Immortalized Human Cells
- Immortalized Murine Cells
- Cell Immortalization Kit
- Adipose Cells
- Cardiac Cells
- Dermal Cells
- Epidermal Cells
- Peripheral Blood Mononuclear Cells
- Umbilical Cord Cells
- Monkey Primary Cells
- Mouse Primary Cells
- Breast Tumor Cells
- Colorectal Tumor Cells
- Esophageal Tumor Cells
- Lung Tumor Cells
- Leukemia/Lymphoma/Myeloma Cells
- Ovarian Tumor Cells
- Pancreatic Tumor Cells
- Mouse Tumor Cells