JHOS-3
Cat.No.: CSC-C6609J
Species: Homo sapiens (Human)
Source: Lymph Node Metastasis
Morphology: Epithelial-like
Culture Properties: Adherent
- Specification
- Background
- Scientific Data
- Q & A
- Customer Review
Shipping: Dry Ice, Frozen
JHOS-3 is a thoroughly described human ovarian serous adenocarcinoma cell line first derived from a Japanese female patient aged between 40 and 50 years. This adherent epithelial cell line exhibits characteristic morphological traits of ovarian cancer and demonstrates modest growth in vitro. JHOS-3 has an aneuploid karyotype with distinct genetic modifications associated with ovarian cancer and is tumorigenic in nude mice, preserving essential pathological features of primary ovarian serous carcinoma. It expresses the established ovarian cancer biomarker CA125 and is extensively utilized in research concerning ovarian cancer proliferation, invasion, metastasis, and chemoresistance. It is cultivated in DMEM/F12 media enriched with 15% fetal bovine serum and non-essential amino acids, facilitating steady passage and experimental modification. This cell line functions as a dependable in vitro model for assessing anticancer agents such as cisplatin, paclitaxel, and PARP inhibitors, in addition to exploring oncogenic signaling pathways including PI3K/Akt, MAPK, and p53, thereby offering significant insights into the molecular mechanisms and therapeutic strategies for human ovarian cancer.
Repurposing Celecoxib for Ovarian Cancer Treatment by Targeting Survivin Signaling
The most common type of reproductive cancer that kills women is ovarian cancer. With a five-year mortality rate just above 45% and high rates of chemo-toxicity and chemo-resistance, it is important to find new treatments that work better and are less harmful. By using ten different types of ovarian cancer cell lines from different histologies, Chuwa et al. wanted to find out if the COX-2 inhibitor celecoxib could help fight tumors.
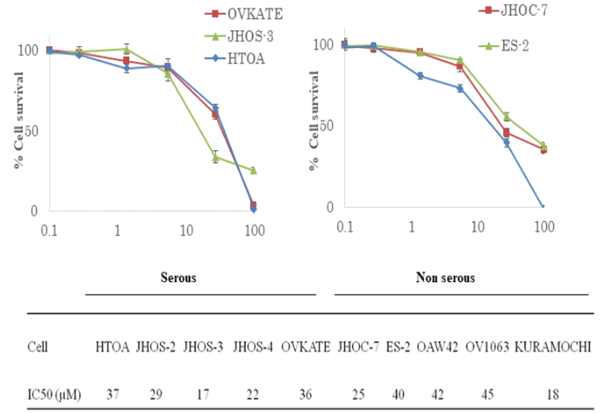
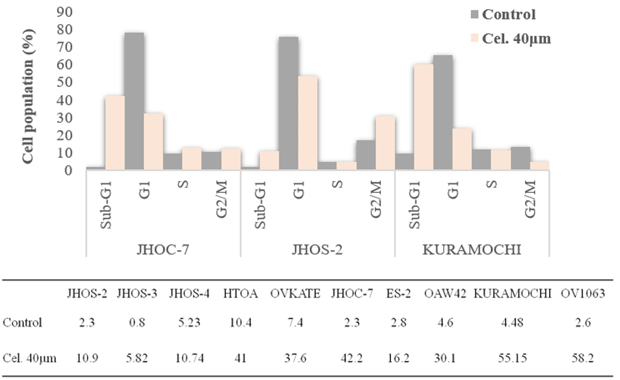
They discovered that celecoxib can inhibit the growth of all the ovarian cancer cell lines (HTOA, JHOC-7, JHOS-2, JHOS-3, and JHOS-4), which derived from different types of tissue. As shown in Fig. 1, celecoxib effectively and significantly slowed the growth of ovarian cancer cells at IC50 values range from 17µm to 45µm. It has already talked about the genetic changes found in ovarian cancer cell lines. Celecoxib's ability to stop cell growth wasn't linked to or changed by the histological traits or mutational states of these cells. They investigated celecoxib's effect on cell cycle development to learn more about how it inhibits the growth of many types of ovarian cancer cells, no matter what the histology is. The picture in Fig. 2 shows that celecoxib changes the cell cycle by significantly increasing the number of sub-G1 cells (p<0.001, paired t-test) in all cell lines. There was no link between celecoxib's effect on the cell cycle, defects, or the structure of the cells, which suggests that the drug has more than one function.
Ask a Question
Write your own review
- You May Also Need
- Adipose Tissue-Derived Stem Cells
- Human Neurons
- Mouse Probe
- Whole Chromosome Painting Probes
- Hepatic Cells
- Renal Cells
- In Vitro ADME Kits
- Tissue Microarray
- Tissue Blocks
- Tissue Sections
- FFPE Cell Pellet
- Probe
- Centromere Probes
- Telomere Probes
- Satellite Enumeration Probes
- Subtelomere Specific Probes
- Bacterial Probes
- ISH/FISH Probes
- Exosome Isolation Kit
- Human Adult Stem Cells
- Mouse Stem Cells
- iPSCs
- Mouse Embryonic Stem Cells
- iPSC Differentiation Kits
- Mesenchymal Stem Cells
- Immortalized Human Cells
- Immortalized Murine Cells
- Cell Immortalization Kit
- Adipose Cells
- Cardiac Cells
- Dermal Cells
- Epidermal Cells
- Peripheral Blood Mononuclear Cells
- Umbilical Cord Cells
- Monkey Primary Cells
- Mouse Primary Cells
- Breast Tumor Cells
- Colorectal Tumor Cells
- Esophageal Tumor Cells
- Lung Tumor Cells
- Leukemia/Lymphoma/Myeloma Cells
- Ovarian Tumor Cells
- Pancreatic Tumor Cells
- Mouse Tumor Cells