Immortalized Rat Retinal Precursor Cells (R28)
Cat.No.: CSC-I9196L
Species: Rattus norvegicus
Source: Retinal Tissue
Culture Properties: Adherent
- Specification
- Background
- Scientific Data
- Q & A
- Customer Review
Note: Never can cells be kept at -20 °C.
2) RT-PCR and immunocytochemistry were used to assess the expression of the photoreceptor specific marker S-antigen and interphotoreceptor retinoid binding protein (IRBP)
The Immortalized Rat Retinal Precursor Cell Line (R28) represents a landmark achievement in ocular cell biology, offering researchers a uniquely powerful and reproducible in vitro tool for the study of retinal physiology, pathology, and pharmacology. Originally established from postnatal day 6 (P6) Sprague-Dawley rat retinal tissue through immortalization with the 12S E1A gene of adenovirus delivered via a replication-incompetent retroviral vector, R28 cells were designed to overcome the significant limitations of previously available retinal cell models.
Unlike retinoblastoma-derived cell lines (e.g., Y79) or SV40-immortalized cultures, R28 cells exhibit contact-inhibited, anchorage-dependent growth and are confirmed to be non-tumorigenic - a critical safety and physiological relevance advantage validated by in vivo transplantation studies. This makes R28 cells far more representative of normal retinal cell behavior than oncogenically transformed alternatives.
A defining feature of R28 cells is their dual neuronal and glial identity, co-expressing key retinal markers including recoverin, interphotoreceptor retinoid-binding protein (IRBP), glial fibrillary acidic protein (GFAP), vimentin, and S-100. This phenotypic heterogeneity closely recapitulates the cellular diversity of the intact retina, enabling broader experimental applicability. Crucially, R28 cells possess functional neurotransmitter receptors - including ionotropic glutamate receptors (GluR1/2/3, NMDA), GABA_A/B receptors, and receptors for dopamine, serotonin, glycine, and acetylcholine - making them uniquely suited for electrophysiological and pharmacological investigations.
Inhibition of Mettl3-Mediated m6A RNA Modification of HMGCS1 Protects Retinal Ganglion Cells in A Rat Model of Glaucoma from Glutamate Excitotoxicity-Induced Ferroptosis
Glaucoma, a leading cause of irreversible blindness worldwide, is characterized by progressive retinal ganglion cells (RGCs) degeneration. The accumulation of glutamate in the retina is a common mechanism underlying the of RGCs death in various forms of glaucoma. Neuroprotective strategies for RGCs remain an unmet need, urging exploration of novel molecular mechanisms beyond intraocular pressure (IOP) control.
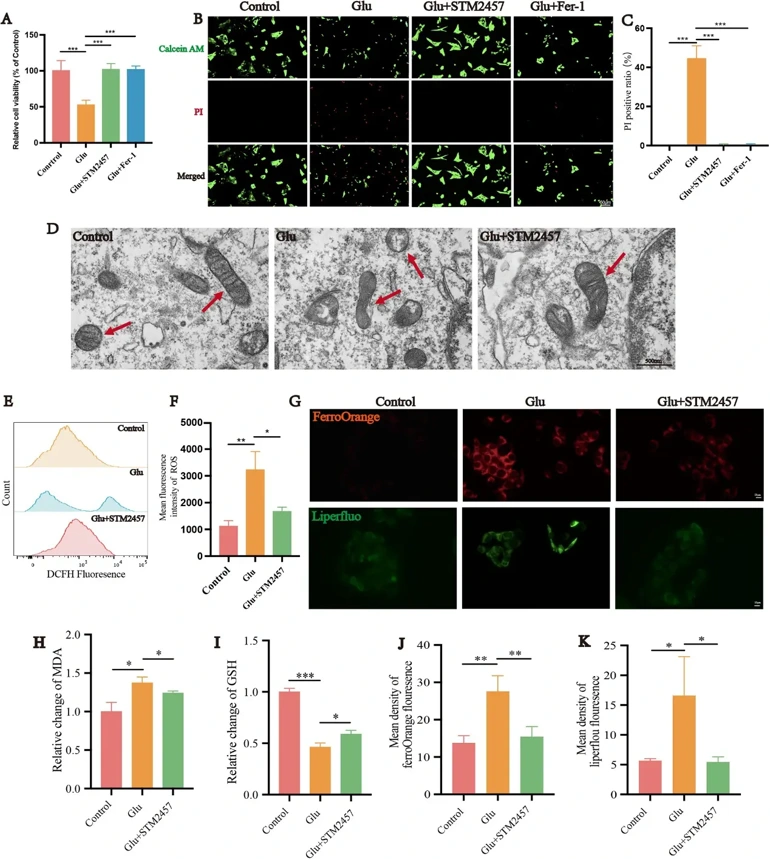
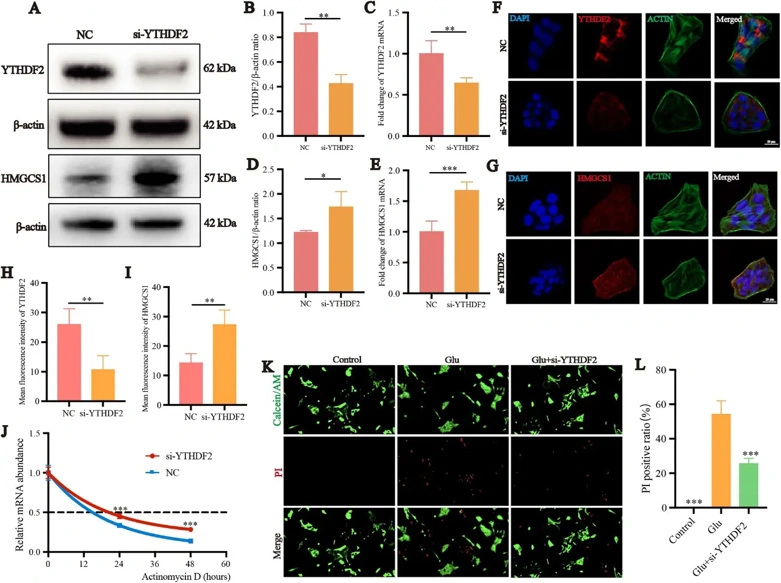
Retinal m6A methylation sequencing, RT-qPCR, and dot blot techniques were performed to identify m6A methylation levels change in an N-methyl-D-aspartate (NMDA)-induced glaucoma model of rats and find potential targets. The effect of methyltransferase-like 3 (Mettl3) inhibition on ferroptosis of R28 and RGCs glutamate excitotoxicity model was detected by using Mettl3 inhibitor STM2457. Using si-RNA and oe-RNA to knockdown YTHN6-methyladenosine RNA binding protein 2 (YTHDF2) or Hydroxyl-3-methylglutaryl-Coenzyme A synthase 1 (HMGCS1) in the R28. Lentiviral vectors were applied to overexpress the HMGCS1 in rat retina. MeRIP-qPCR and Western Blot to study the mechanism of how Mettl3 regulates m6A methylation and expression of downstream targets.
The findings demonstrate that NMDA-induced excitotoxicity significantly elevated retinal m6A methylation levels. HMGCS1 m6A methylation was significantly increased while its expression significantly decreased in the NMDA group. In R28 cells, inhibition of Mettl3 significantly alleviated glutamate excitotoxicity-induced R28 and RGCs ferroptosis and restored the visual function of rats. Knockdown HMGCS1 significantly reduced the protective effect of Mettl3 inhibition on the R28 cells and overexpress HMGCS1 protected R28 cell and RGCs from NMDA-induced glutamate excitotoxicity. YTHDF2 reverses this protective effect by recognizing and degrading m6A-modified HMGCS1 mRNA, thereby promoting ferroptosis.
Ask a Question
Write your own review
- Adipose Tissue-Derived Stem Cells
- Human Neurons
- Mouse Probe
- Whole Chromosome Painting Probes
- Hepatic Cells
- Renal Cells
- In Vitro ADME Kits
- Tissue Microarray
- Tissue Blocks
- Tissue Sections
- FFPE Cell Pellet
- Probe
- Centromere Probes
- Telomere Probes
- Satellite Enumeration Probes
- Subtelomere Specific Probes
- Bacterial Probes
- ISH/FISH Probes
- Exosome Isolation Kit
- Human Adult Stem Cells
- Mouse Stem Cells
- iPSCs
- Mouse Embryonic Stem Cells
- iPSC Differentiation Kits
- Mesenchymal Stem Cells
- Immortalized Human Cells
- Immortalized Murine Cells
- Cell Immortalization Kit
- Adipose Cells
- Cardiac Cells
- Dermal Cells
- Epidermal Cells
- Peripheral Blood Mononuclear Cells
- Umbilical Cord Cells
- Monkey Primary Cells
- Mouse Primary Cells
- Breast Tumor Cells
- Colorectal Tumor Cells
- Esophageal Tumor Cells
- Lung Tumor Cells
- Leukemia/Lymphoma/Myeloma Cells
- Ovarian Tumor Cells
- Pancreatic Tumor Cells
- Mouse Tumor Cells