PA317
Cat.No.: CSC-C9121W
Species: Mus musculus (Mouse)
Source: Embryo
Morphology: fibroblast
- Specification
- Background
- Scientific Data
- Q & A
- Customer Review
PA317 is an amphotropic retrovirus packaging line commonly used in laboratory research settings. The line was created by Miller and Buttimore in 1986 as a derivative of mouse embryo fibroblast line NIH 3T3. This was achieved by co-transfecting both pPAM3 (the retroviral packaging construct) and the herpes simplex virus thymidine kinase (TK) gene into NIH 3T3 cells. The primary innovation of this line lies in its genomic modifications: the viral packaging signal (psi) was deleted and the 3' Long Terminal Repeat (LTR) was partially replaced. This causes cells to express necessary proteins for viral packaging in trans while greatly decreasing chances of replication competent helper viruses forming from recombination.
Cells of this line are characterized by their amphotropic viral host range. In contrast to ecotropic packaging lines which only generate viruses able to infect rodent cells, viruses produced from PA317 infected cells can infect target cells of many different species including humans, birds, and cats. For this reason, PA317 was one of the first packaging lines used for research into gene therapy and was used to deliver gene therapy constructs in phase I clinical trials.
Anti-Glycative and Anti-Inflammatory Effects of Macamides Isolated from Tropaeolum tuberosum in Skin Cells
Tropaeolum tuberosum, commonly known as Mashua, is an herbal remedy used on the skin in order to treat local pain and to heal wounds. Ticona et al. aimed to evaluate the extracts and isolated compounds from T. tuberosum with anti-glycative and anti-inflammatory activities.
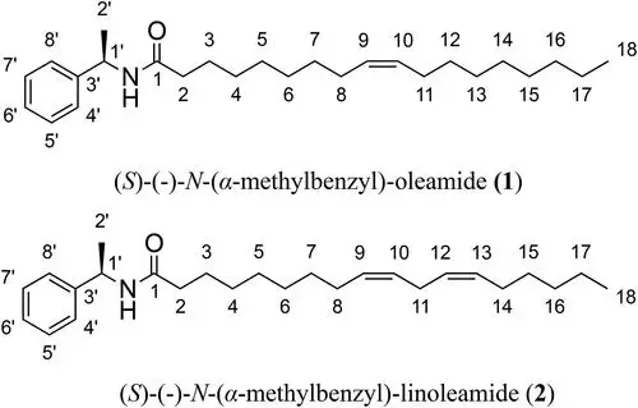
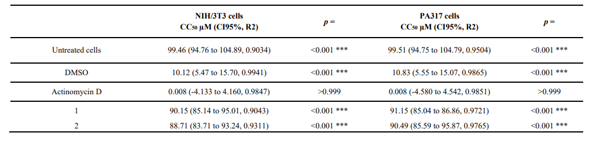
The isolated compounds were identified as (S)-(-)-N-(α-methylbenzyl)-oleamide (1) and (S)-(-)-N-(α-methylbenzyl)-linoleamide (2) (Fig. 1). These compounds were previously described by D'Oca et al. Their cytotoxicity was assessed using the XTT assay in NIH/3T3 and PA317 cells for 12 hours. The results showed that neither compound significantly reduced cell viability. The CC50 values for compound 1 were 90.15 µM (NIH/3T3) and 91.15 µM (PA317), while for compound 2, they were 88.71 µM (NIH/3T3) and 90.49 µM (PA317), all higher than the positive control actinomycin D (ACT, CC50 = 0.008 µM). This indicates that compounds 1 and 2 were less cytotoxic than ACT. The lower cytotoxicity of compounds 1 and 2 is attributed to their increased lipophilicity due to the presence of double bonds in the side chain, confirming the relationship between cytotoxicity and lipophilic capacity. Lipophilicity is measured as LogP, where higher LogP values enhance cell permeability. Compound 2 had a cLogP of 8.20, allowing it to cross cell membranes more easily than compound 1 (cLogP of 7.22). Thus, compounds with more double bonds in their side chains can more readily diffuse across cell barriers.
Ask a Question
Write your own review
- Adipose Tissue-Derived Stem Cells
- Human Neurons
- Mouse Probe
- Whole Chromosome Painting Probes
- Hepatic Cells
- Renal Cells
- In Vitro ADME Kits
- Tissue Microarray
- Tissue Blocks
- Tissue Sections
- FFPE Cell Pellet
- Probe
- Centromere Probes
- Telomere Probes
- Satellite Enumeration Probes
- Subtelomere Specific Probes
- Bacterial Probes
- ISH/FISH Probes
- Exosome Isolation Kit
- Human Adult Stem Cells
- Mouse Stem Cells
- iPSCs
- Mouse Embryonic Stem Cells
- iPSC Differentiation Kits
- Mesenchymal Stem Cells
- Immortalized Human Cells
- Immortalized Murine Cells
- Cell Immortalization Kit
- Adipose Cells
- Cardiac Cells
- Dermal Cells
- Epidermal Cells
- Peripheral Blood Mononuclear Cells
- Umbilical Cord Cells
- Monkey Primary Cells
- Mouse Primary Cells
- Breast Tumor Cells
- Colorectal Tumor Cells
- Esophageal Tumor Cells
- Lung Tumor Cells
- Leukemia/Lymphoma/Myeloma Cells
- Ovarian Tumor Cells
- Pancreatic Tumor Cells
- Mouse Tumor Cells