KYSE-30
Cat.No.: CSC-C0405
Species: Homo sapiens (Human)
Source: Esophagus
Morphology: adherent, epitheloid with long processes growing in monolayers
Culture Properties: monolayer
- Specification
- Background
- Scientific Data
- Q & A
- Customer Review
Immunology: cytokeratin +, cytokeratin-7 -, cytokeratin-8 +, cytokeratin-17 -, cytokeratin-18 +, cy
Derived from a well-differentiated invasive esophageal squamous cell carcinoma resected from a 64-year-old Asian male prior to treatment, KYSE-30 is a prototypic human ESCC line established via initial heterotransplantation in athymic mice. Its extensively characterized genomic landscape and stable phenotype position it as a cornerstone model for dissecting ESCC pathogenesis and therapeutic response.
KYSE-30 harbors a pathogenic TP53 splice-site mutation (intron 6) alongside CDKN2A and HRAS mutations, coupled with high-level co-amplification of MYC, ERBB2 (c-ERB B), and CCND1. This combinatorial genotype faithfully recapitulates the core oncogenic circuitry prevalent in human ESCC.
Genomic profiling reveals that cell cycle gene mutations occur at low frequency across ESCC lines; KYSE-30 is one of only two lines (with TE-1) exhibiting this distinct mutational class. This exclusivity renders it indispensable for studying G1/S checkpoint abrogation and CDK inhibitor sensitivity in an ESCC-relevant context.
KYSE-30 expresses an exceptionally high level of epidermal growth factor receptor, providing a calibrated system for interrogating EGFR-dependent signaling, tyrosine kinase inhibitor efficacy, and receptor-targeted therapeutic strategies.
The line is heterotransplantable in nude mice, enabling in vivo efficacy studies. Engineered derivatives, including GFP-reporter stable lines, support high-content drug screening and real-time tumor tracking in preclinical models.
CAP1 Signaling Regulates Tumor Immunity, Proliferation, and Migration in Esophageal Cancer
Esophageal cancer (ESCA) is a common malignancy with high morbidity and mortality. Our study aimed to explore the regulatory pathways of CAP1 in ESCA and its effects on tumor immunity.
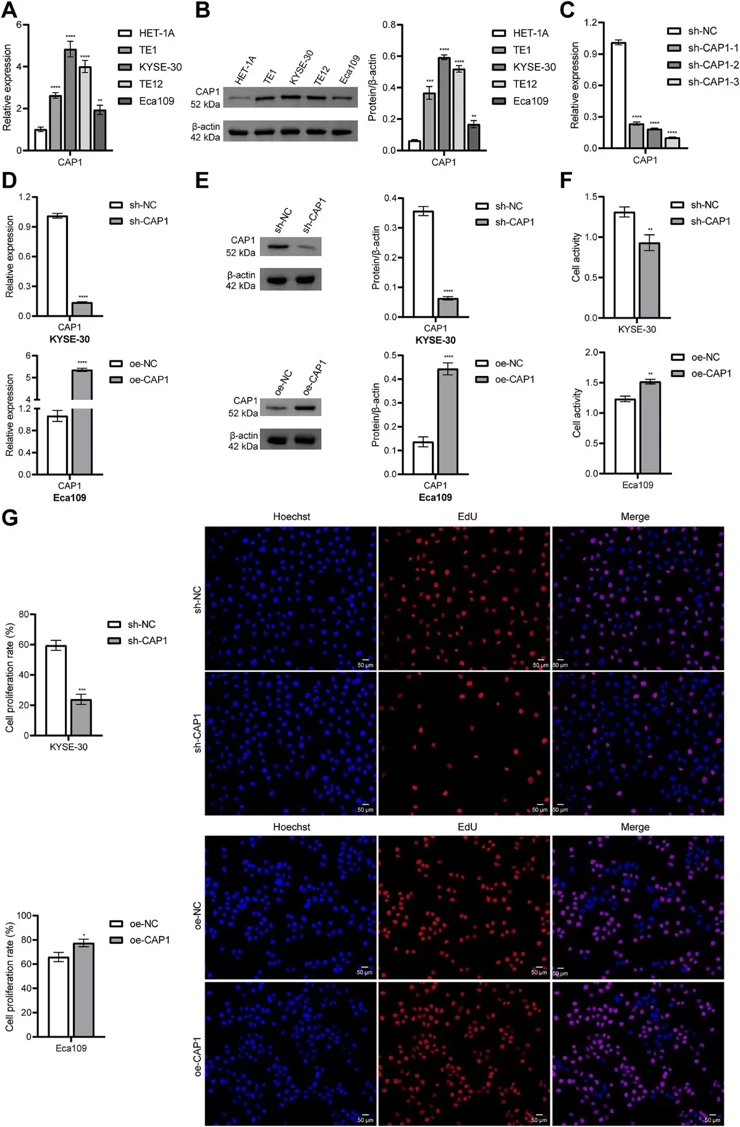
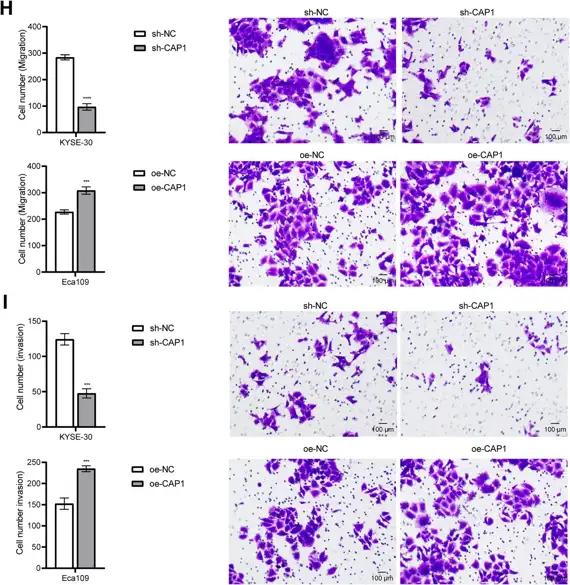
Bioinformatics analysis using LASSO Cox regression demonstrated that CAP1 carried the highest risk coefficient (0.4437) in the prognostic model. The risk score model demonstrated excellent predictive performance in the TCGA cohort and was successfully validated in external GEO datasets. High expression of CAP1 in ESCA cells enhanced their proliferative, migratory, and invasive capabilities. In a co-culture system, silencing CAP1 in ESCA cells enhanced the cytotoxicity of CD8+ T cells and reduced their apoptosis. The transcription factor ELF1 promoted CAP1 expression by binding to its promoter, thereby regulating immune evasion and tumor progression in ESCA. Importantly, in vivo experiments confirmed that knockdown of either ELF1 or CAP1 significantly suppressed tumor growth, enhanced CD8+ T cell infiltration, and downregulated PD-L1 expression, demonstrating that ELF1 promotes tumor progression and immune escape in ESCA via CAP1.
Ask a Question
Write your own review
- You May Also Need
- Adipose Tissue-Derived Stem Cells
- Human Neurons
- Mouse Probe
- Whole Chromosome Painting Probes
- Hepatic Cells
- Renal Cells
- In Vitro ADME Kits
- Tissue Microarray
- Tissue Blocks
- Tissue Sections
- FFPE Cell Pellet
- Probe
- Centromere Probes
- Telomere Probes
- Satellite Enumeration Probes
- Subtelomere Specific Probes
- Bacterial Probes
- ISH/FISH Probes
- Exosome Isolation Kit
- Human Adult Stem Cells
- Mouse Stem Cells
- iPSCs
- Mouse Embryonic Stem Cells
- iPSC Differentiation Kits
- Mesenchymal Stem Cells
- Immortalized Human Cells
- Immortalized Murine Cells
- Cell Immortalization Kit
- Adipose Cells
- Cardiac Cells
- Dermal Cells
- Epidermal Cells
- Peripheral Blood Mononuclear Cells
- Umbilical Cord Cells
- Monkey Primary Cells
- Mouse Primary Cells
- Breast Tumor Cells
- Colorectal Tumor Cells
- Esophageal Tumor Cells
- Lung Tumor Cells
- Leukemia/Lymphoma/Myeloma Cells
- Ovarian Tumor Cells
- Pancreatic Tumor Cells
- Mouse Tumor Cells





