Mouse Dental Pulp Stem Cells
- Specification
- Background
- Scientific Data
- Q & A
- Customer Review
Mouse Dental Pulp Stem Cells (mDPSCs) are a distinct population of mesenchymal stem cells (MSCs) harvested from the soft vital tissue within the dental pulp chamber. Recognized for their remarkable embryological origin-the cranial neural crest-mDPSCs possess unique neuro-ectodermal properties that distinguish them from traditional bone marrow-derived MSCs. This makes them a premier biological resource for regenerative medicine, dental tissue engineering, and systemic therapeutic modeling.
- Exceptional Proliferative Potency: mDPSCs exhibit a higher proliferation rate and a more robust colony-forming efficiency compared to many other adult stem cell sources. This high mitotic activity allows for rapid ex vivo expansion, facilitating the large-scale production required for industrial and pharmaceutical applications.
- Enhanced Multilineage Plasticity: Beyond standard tri-lineage differentiation (osteogenic, chondrogenic, and adipogenic), mDPSCs demonstrate superior neurogenic and odontogenic potential. Their neural crest origin enables them to differentiate into functional neurons and glia, making them an ideal model for neurodegenerative disease research.
- Robust Immunomodulatory & Paracrine Functions: mDPSCs secrete a diverse array of bioactive molecules, including VEGF, BDNF, and GDNF. These secretomes play a critical role in modulating immune responses and promoting angiogenesis, which are essential for repairing damaged tissues and treating inflammatory disorders.
- Accessible & Ethical Modeling: In the context of preclinical research, mouse-derived DPSCs provide an invaluable tool for utilizing transgenic and knockout mouse models. This allows for the precise study of gene function in stem cell niche maintenance and tissue regeneration before clinical translation.
Our Mouse Dental Pulp Stem Cells undergo stringent validation to ensure the retention of MSC surface markers (CD73, CD90, CD105) and multi-potentiality. By providing a standardized, high-performance cell system, this product serves as an essential platform for researchers aiming to accelerate discoveries in stem cell biology, craniofacial reconstruction, and cell-based drug delivery systems.
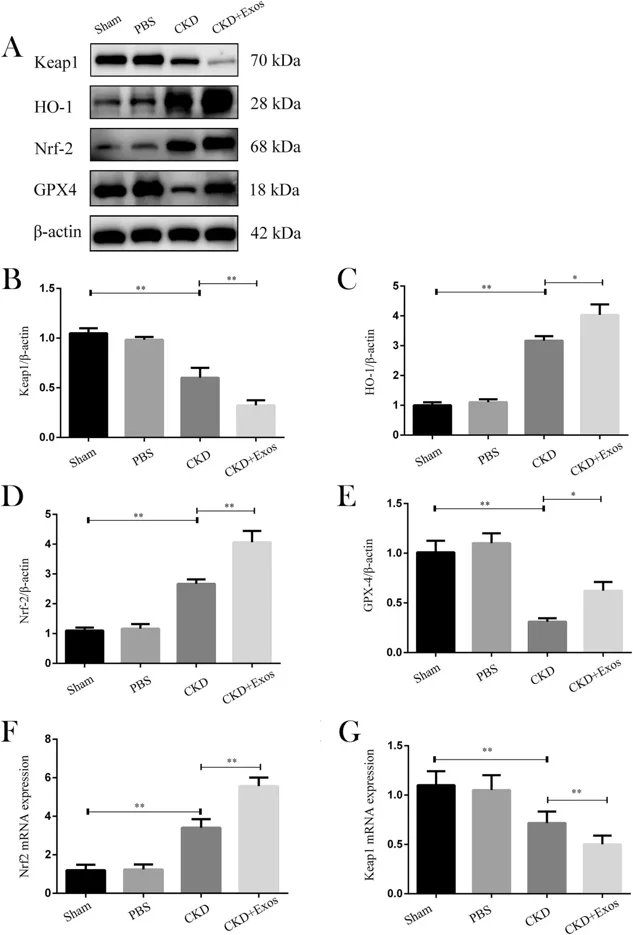
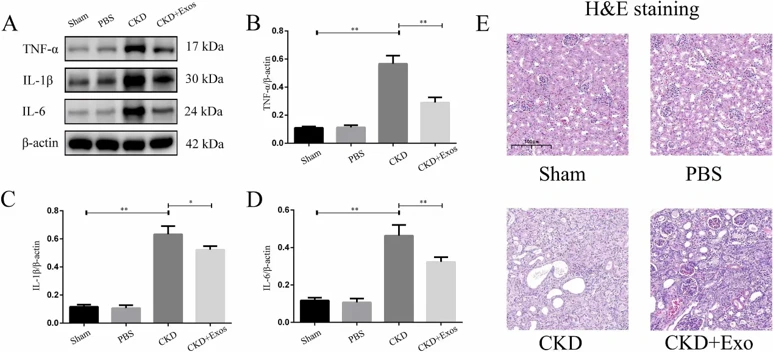
Dental Pulp Stem Cells Derived Exosomes Inhibit Ferroptosis to Ameliorate Chronic Kidney Disease Injury
Chronic kidney disease (CKD) has long represented a substantial global health challenge. Regrettably, current therapeutic interventions exhibit limited efficacy in halting the progression of CKD. Ferroptosis may play a crucial role in CKD, as indicated by substantial evidence. Dental pulp stem cell-derived exosomes (DPSC-Exos) possess advantages such as abundant sources and low immunogenicity, holding promising prospects in CKD treatment.
This study constructed a mouse CKD model to investigate the therapeutic effects of DPSC-Exos. First, we successfully extracted and identified DPSC-Exos. Then, mice were randomly divided into sham, PBS, CKD, and CKD+Exos groups. Our study determined the expression of ferroptosis-related pathway molecules Nrf2, GPX4, Keap1, and HO-1 in each group. Finally, we detected the expression levels of inflammatory factors, TNF-α, IL-1β, and IL-6, at the injury site.
Mice treated with DPSC-Exos showed increased expression of the ferroptosis inhibitory factor Nrf2 and its downstream regulatory factors GPX4 and HO-1, while the expression of Keap1 decreased. The expression of TNF-α, IL-1β, and IL-6 also decreased. DPSC-Exos may help inhibit ferroptosis through the Keap1-Nrf2/GPX4 pathway and reduce the inflammatory response at the injury site, revealing their potential therapeutic effects on CKD.
Ask a Question
Write your own review
- Adipose Tissue-Derived Stem Cells
- Human Neurons
- Mouse Probe
- Whole Chromosome Painting Probes
- Hepatic Cells
- Renal Cells
- In Vitro ADME Kits
- Tissue Microarray
- Tissue Blocks
- Tissue Sections
- FFPE Cell Pellet
- Probe
- Centromere Probes
- Telomere Probes
- Satellite Enumeration Probes
- Subtelomere Specific Probes
- Bacterial Probes
- ISH/FISH Probes
- Exosome Isolation Kit
- Human Adult Stem Cells
- Mouse Stem Cells
- iPSCs
- Mouse Embryonic Stem Cells
- iPSC Differentiation Kits
- Mesenchymal Stem Cells
- Immortalized Human Cells
- Immortalized Murine Cells
- Cell Immortalization Kit
- Adipose Cells
- Cardiac Cells
- Dermal Cells
- Epidermal Cells
- Peripheral Blood Mononuclear Cells
- Umbilical Cord Cells
- Monkey Primary Cells
- Mouse Primary Cells
- Breast Tumor Cells
- Colorectal Tumor Cells
- Esophageal Tumor Cells
- Lung Tumor Cells
- Leukemia/Lymphoma/Myeloma Cells
- Ovarian Tumor Cells
- Pancreatic Tumor Cells
- Mouse Tumor Cells