- Specification
- Background
- Scientific Data
- Q & A
- Customer Review
These adult melanocyte cultures are available at 95% purity and are recommended with optimized media formulated specifically to support growth and survival in vitro.
Human adult melanocytes are pigment-producing cells primarily localized in the basal layer of the epidermis, where they form functional units with adjacent keratinocytes via dendritic processes. Derived from neural crest progenitors, these terminally differentiated cells exhibit remarkable stability and retain their hallmark functions throughout adulthood. Their principal physiological role is the synthesis of melanin within specialized organelles termed melanosomes, followed by transfer to keratinocytes, providing essential photoprotection against ultraviolet radiation (UVR)-induced DNA damage.
A key advantage of human adult melanocytes lies in their phenotypic and functional authenticity compared to immortalized cell lines or neonatal/embryonic counterparts. They maintain endogenous expression of melanocyte-specific markers, including microphthalmia-associated transcription factor (MITF), tyrosinase (TYR), tyrosinase-related protein 1 (TRP1/TYRP1) and TRP2 (DCT), alongside characteristic dendritic morphology. From a translational perspective, primary human adult melanocytes offer a physiologically relevant ex vivo model for investigating pigmentation disorders (e.g., vitiligo, melasma), melanoma pathogenesis, and drug or cosmetic compound screening. Their availability from different skin types and phototypes enables the study of ethnic variation in pigmentation. Importantly, these cells respond to hormonal and paracrine signals in a manner recapitulating native tissue behavior, rendering them indispensable for mechanistic studies and safety assessments in dermatological research.
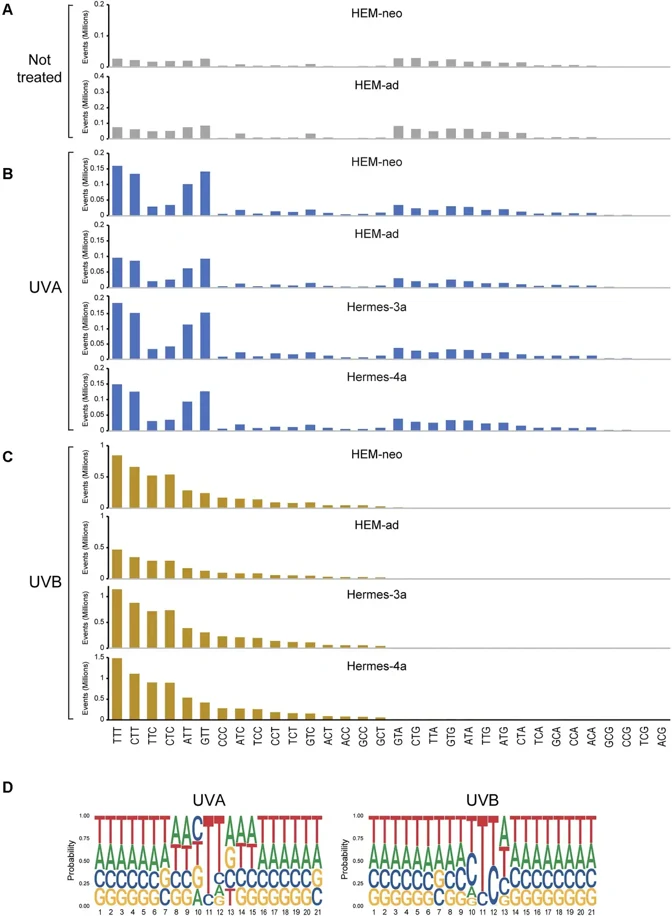
UVA-Induced DNA Damage and Mutations in Human Melanocytes
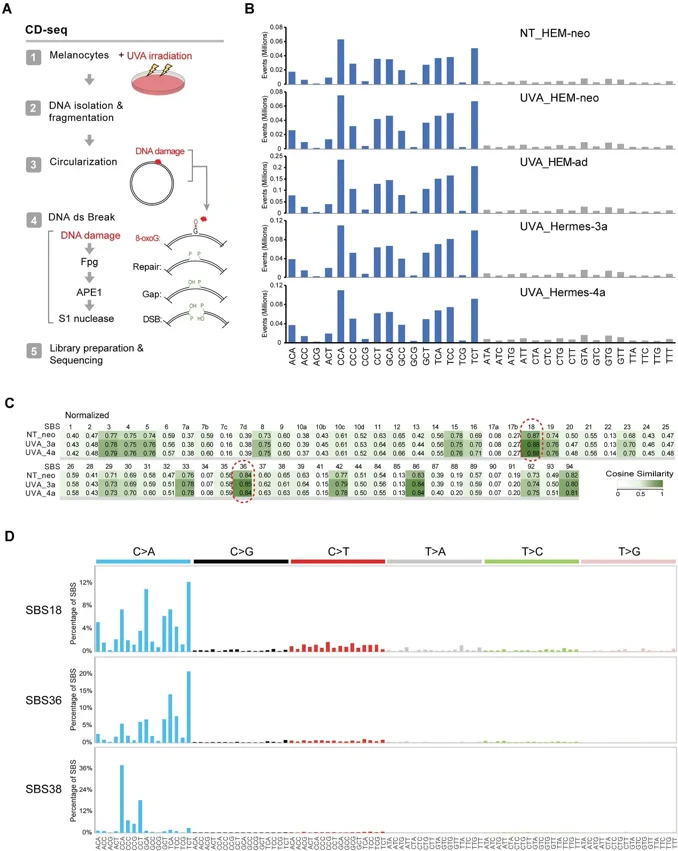
UVB radiation (280-320 nm) from sunlight induces skin cancer through DNA damage-induced mutations. Melanomas carry mutational signatures associated with UVB-induced cyclobutane pyrimidine dimers (CPDs). However, there are several other melanoma signatures of unknown origin. To test if these signatures are linked to UVA, we exposed human melanocytes (HEM) to UVA (340-400 nm) and to UVB for comparison. We used five different HEM lines, two neonatal foreskin melanocyte lines (HEM-neo and HEMn-LP), one line of adult human melanocytes (HEM-ad), and the telomerase-immortalized, nontransformed melanocyte lines Hemes-3a and Hermes-4a.
We mapped DNA damage in the form of CPDs or 8-oxoguanines (8-oxoG) genome-wide at base resolution. We then determined mutational patterns in single melanocyte cell clones by whole genome sequencing. Different from UVB, UVA induces CPDs more selectively at TT sequences resembling melanoma signature SBS7d. We did not observe rising CPD levels after cessation of radiation (dark CPDs). The UVA-induced CPDs were not mutagenic in the mutation analysis. 8-oxoG was present in melanocytes but did not substantially increase after UVA. G/C to T/A mutations were prominent in melanocyte single-cell clones with no major shift after UVA radiation. These mutations matched SBS18, a signature present in melanomas. Although UVA damages DNA, it has a surprisingly limited mutagenic effect on human melanocytes. However, the oxidative base lesions in melanocytes and their associated mutations may be linked to a subset of melanoma mutations.
The cells have been tested for melanogenesis activity by immunofluorescent labeling of Mel-5 and L-dopa conversion activity. We don’t have a particular listing of each lot’s melanogenesis activity, however, each lot is tested for L-DOPA conversion as a QC measure of their capability to synthesize melanin. All of the lots that I have listed have passed our QC for Mel-5 positivity (>80%) and have been stained for L-DOPA conversion (but not quantified). We can provide the CoA for the specific lot that you will order.
Ask a Question
Write your own review