Rat Cerebellar Granule Cells
Cat.No.: CSC-C1786
Species: Rat
Source: Brain
Cell Type: Granule Cell
- Specification
- Background
- Scientific Data
- Q & A
- Customer Review
RCGC are isolated from neonate day 8 rat cerebellum. RCGC are cryopreserved at primary culture and delivered frozen. Each vial contains >1 x 10^6 cells in 1 ml volume. RCGC are characterized by immunofluorescent method with antibodies to neurafilament, MAP2, and beta-tubulin 3. RCGC are negative for mycoplasma, bacteria, yeast and fungi. RCGC are guaranteed to further culture in the conditions provided by Creative Bioarray.
Rat Cerebellar Granule Cells, commonly abbreviated as RCGCs, are neuronal cells isolated from cerebellum of newborn rats. RCGCs are one of the most commonly used primary neuronal cell cultures in research. Granule cells are glutamatergic neurons that account for approximately two-thirds of all cells in the cerebellum. They are often used in studies examining neurotransmission, neuronal differentiation, and signal transduction. Rat cerebellar granule cells are often chosen due to the relative homogeneity of the cell preparation and their known biological properties.
Primary Rat cerebellar granule cells cultured in vitro have been shown to have extensive neurite outgrowth, and express neuronal markers such as NeuN, MAP2, and synaptophysin. RCGCs have also been shown to be sensitive to alterations in potassium concentrations, oxidative stress, and excitotoxicity. Because of this sensitivity they are often used as a model to study neuronal cell death and survival. Rat cerebellar granule cells have been used to study glutamate receptors as well as stroke. RCGCs are also commonly used for testing candidate drugs for neuroprotective or neurotoxic effects.
Involvement Of GABAA Receptors Containing α6 Subtypes in Antisecretory Factor Activity on Rat Cerebellar Granule Cells Studied by Two-Photon Uncaging
Antisecretory factor (AF) is an endogenous protein that counteracts intestinal hypersecretion and inflammation, but its mechanisms are unclear. Bazzurro et al. investigated AF's pharmacological effects on different GABAA receptor populations in cerebellar granule cells using the two-photon uncaging method.
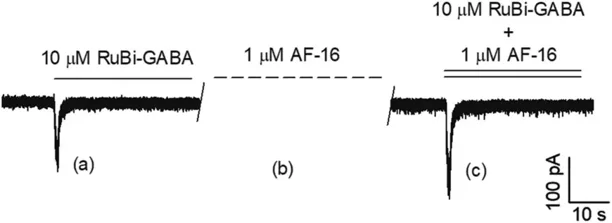
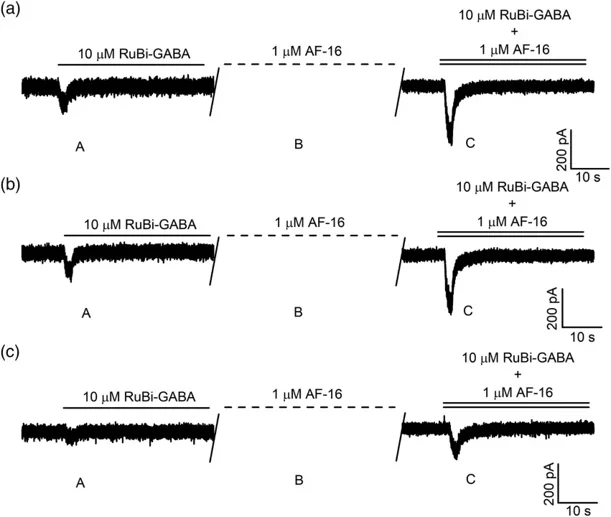
To evaluate the effects of AF-16 on specific neuronal regions, they focused on the soma, axon initial segment (AIS), and neurites (Fig. 1). They applied AF-16 at 1.0 μM for 3 minutes, based on previous findings by Bazzurro et al. (2018) that this concentration maximizes the enhancement of the peak current component. Figure 2 illustrates typical chloride currents evoked by RuBi-GABA photolysis using two-photon excitation (2PE) near the soma. During experiments, they continuously measured the current while following this procedure: They perfused 10-μM RuBi-GABA onto CGCs, uncaged it (Fig. 2a), and then washed it away. Next, we applied 1.0-μM AF-16 for 3 minutes (Fig. 2b), followed by a solution of 10-μM RuBi-GABA and 1.0-μM AF-16, and reactivated the 2PE beam (Fig. 2c). They repeated this process in different neuronal regions and reported the typical currents in Figure 3.

Ask a Question
Write your own review