Diabetic Mouse Retinal Microvascular Endothelial Cells
Cat.No.: CSC-C9315J
Species: Mouse
Source: Retina; Eye
Cell Type: Endothelial Cell; Microvascular Cell
- Specification
- Background
- Scientific Data
- Q & A
- Customer Review
Diabetic Mouse Retinal Microvascular Endothelial Cells (RMECs) are primary endothelial cells obtained from mouse retinal microvasculature of diabetes animal models induced by streptozotocin or spontaneous diabetic mice. The cells phenotypically and physiologically mimic characteristics observed in diabetes-related microvascular dysfunction such as diabetic retinopathy.
Morphologically, diabetic mouse RMECs exhibit a typical cobblestone-like endothelial appearance and form confluent monolayers under standard culture conditions. They differ from retinal endothelial cells of non-diabetic animals by displaying a dysfunctional endothelial barrier, disrupted tight junctions, and increased permeability. diabetic mouse RMECs also show altered expression of endothelial cell markers such as vascular endothelial (VE)-cadherin and Cluster of Differentiation 31 (CD31). Moreover, they upregulate pro-inflammatory cytokines, cell adhesion molecules, and pathways associated with oxidative stress.
Endothelial cell inflammation, leukostasis, angiogenesis, and apoptosis are amongst the different mechanisms of diabetic retinopathy that can be studied using diabetic mouse RMECs. In addition, they are utilized to understand hyperglycemia-induced pathways including vascular endothelial growth factor (VEGF), protein kinase C (PKC) and nuclear factor kappa B (NF-κB). Moreover, diabetic mouse RMECs can be used to screen potential anti-angiogenic and vasoprotective drugs.
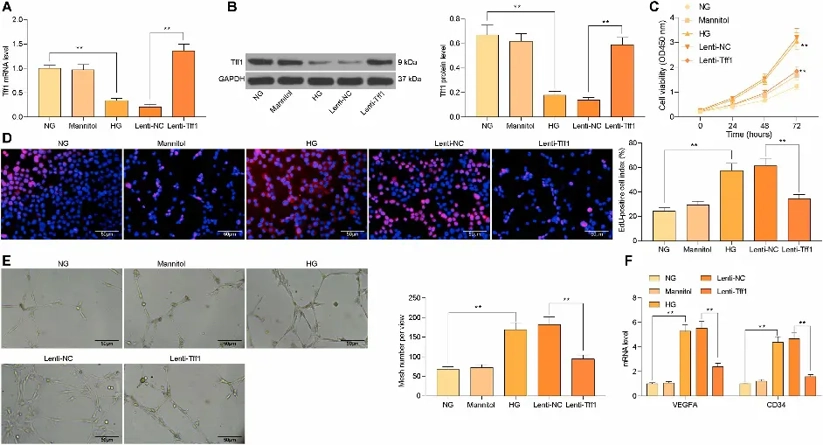
Overexpression of Tff1 Inhibits HG-induced mRMEC Proliferation and Angiogenesis
Diabetic retinopathy (DR) is one of the microvascular complications of diabetes, however, molecular mechanisms of vascular dysfunction, especially endothelial dysfunction in DR are not well understood. Zhang's team developed diabetic retinopathy animal model in mice and cell model in mouse retinal microvascular endothelial cells (mRMECs) to explore the effect of Trefoil factor family 1 (Tff1) in DR.
To understand the function of Tff1 in diabetic retinopathy (DR)-associated endothelial dysfunction, mRMECs were exposed to high glucose (HG, 25 mM). The results showed that HG markedly decreased Tff1 expression, therefore, Tff1 was overexpressed followed by efficiency tested by RT-qPCR and western blot (Fig. 1A, B). Then CCK-8 and EdU assays were performed and data showed that proliferation caused by HG was significantly inhibited by Tff1 overexpression (Fig. 1C, D). Consistently, tube formation assay results showed that angiogenesis promoted by HG was also attenuated after upregulation of Tff1 (Fig. 1E). Moreover, RT-qPCR results showed that overexpression of Tff1 significantly decreased the mRNA expression of VEGFA and CD34 (Fig. 1F). Overall, these data indicated that Tff1 could negatively regulate HG-induced endothelial proliferation and angiogenesis in DR.
Ask a Question
Write your own review
- You May Also Need