C57BL/6 Mouse Bone Marrow Macrophages (Frozen Vial)
Cat.No.: CSC-C4297X
Species: Mouse
Source: Bone Marrow
Cell Type: Macrophage
- Specification
- Background
- Scientific Data
- Q & A
- Customer Review
Mouse Bone Marrow Macrophages are negative for bacteria, yeast, fungi, and mycoplasma. Cells can be expanded on a multiwell culture plate ready for experiments under the cell culture conditions specified by Creative Bioarray. Repeated freezing and thawing of cells is not recommended.
Mouse Bone Marrow Macrophages are tested for expression of markers using antibodies, CD11b by flow cytometry.
Standard biochemical procedures performed with cell cultures include RT-PCR, Western blotting, immunoprecipitation, immunofluorescent staining, flow cytometry or generating cell derivatives for desired research applications.
Bone marrow-derived macrophages (BMDMs) generated from C57BL/6 mice represent the most physiologically relevant and widely adopted primary macrophage model in immunological and pharmaceutical research. Differentiated from bone marrow hematopoietic precursors under M-CSF stimulation, these cells faithfully recapitulate the phenotypic and functional properties of tissue-resident macrophages.
A major advantage of C57BL/6 BMDMs lies in the well-characterized genetic background of the donor strain, which facilitates experimental reproducibility and cross-laboratory comparability. These macrophages retain canonical features, including expression of F4/80, CD11b, and CSF1R, along with potent phagocytic and microbicidal activities. They are exquisitely responsive to polarizing signals (LPS/IFN-γ for M1-like; IL-4/IL-13 for M2-like), enabling mechanistic dissection of macrophage polarization in immunity, inflammation, and tissue remodeling.
Crucially, the C57BL/6 background is permissive for numerous transgenic and knockout lines, allowing straightforward derivation of BMDMs from genetically modified mice. This permits targeted investigation of specific genes or signaling pathways without confounding strain variability. BMDMs are also amenable to CRISPR-Cas9 editing and lentiviral transduction, enhancing their utility for functional genomics. Collectively, C57BL/6 BMDMs serve as a gold-standard ex vivo model for studying macrophage biology, host-pathogen interactions, drug screening, and immunological disorders.
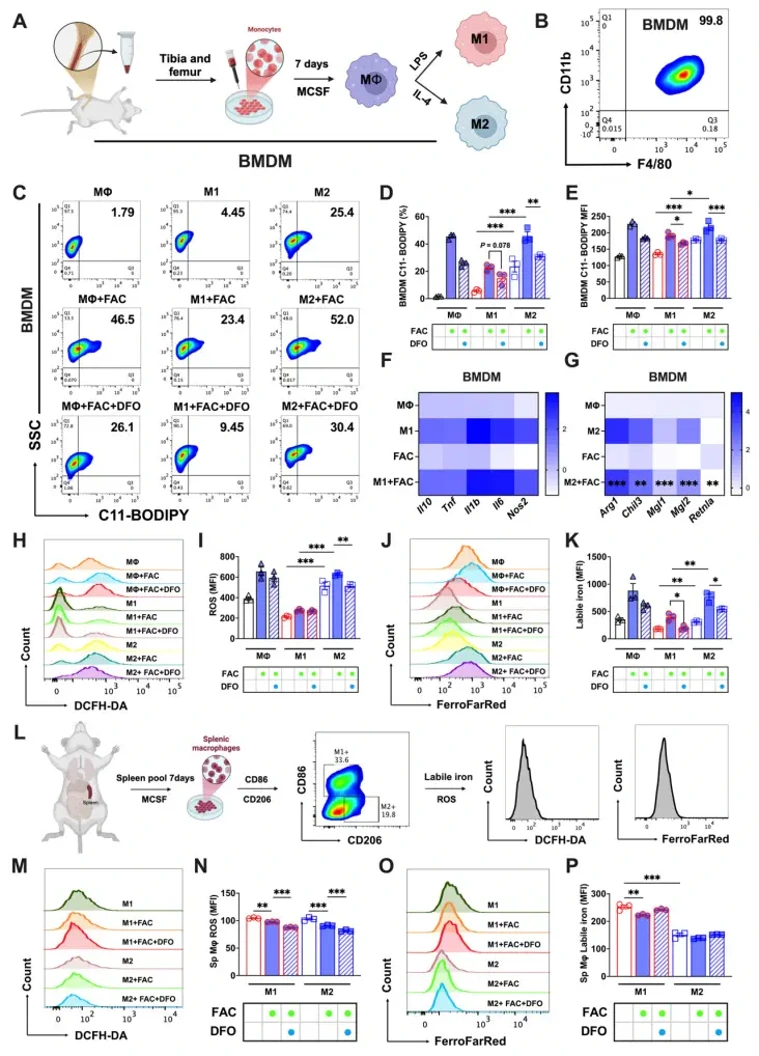
Ferritinophagy Activation States Determine the Susceptibility to Ferroptosis of Macrophages in Bone Marrow and Spleen
Macrophages exhibit heterogeneity due to their presence in different tissues that have distinct cell fates. Ferroptosis is one type of cellular fate, but the sensitivity of different types of macrophages to ferroptosis and the associated molecular mechanisms are not clear. This study explored the ferroptosis sensitivity of bone marrow and splenic macrophage, focusing on the contribution of ferritinophagy. We found that bone marrow M2 macrophages were more susceptible to ferroptosis, which was attributed to their lower solute carrier family 40 member 1 (SLC40A1) and ferritin heavy/light chain (FTH/L) expression and higher labile iron levels compared to those of splenic macrophages. Further, ferritinophagy activation, particularly in M2 macrophages, was identified as the primary cause of increased labile iron levels, as evidenced by experiments using autophagic flux modifiers and RAW264.7 cells with autophagy related 5 (ATG5) and nuclear receptor coactivator 4 (NCOA4) knockdown and NCOA4 knockout. These results provide a new direction for further understanding the heterogeneity and functionality of macrophages, and offers innovative treatments for a variety of health issues in which macrophage regulation plays a critical role.
Ask a Question
Write your own review