- Specification
- Background
- Scientific Data
- Q & A
- Customer Review
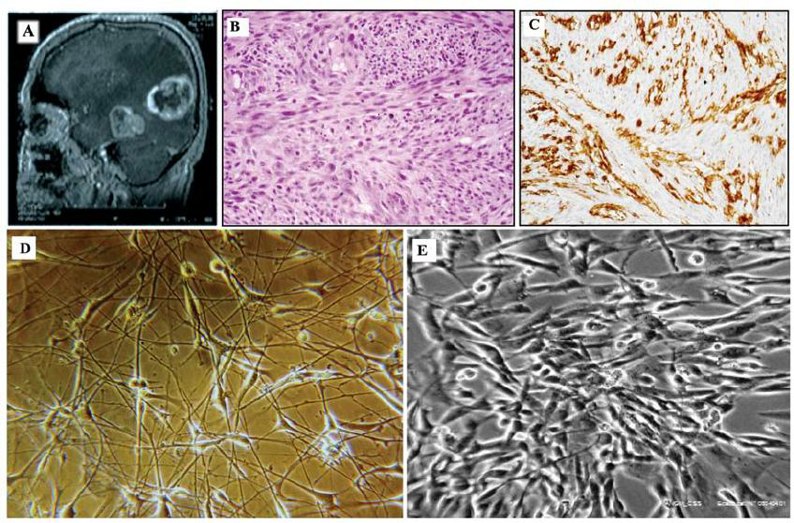
ANGM‑CSS was originally isolated from surgical resection of a left temporo‑parietal GBM from male patient. The resected tumor specimen was cut into 1-2 mm pieces, placed into poly‑D‑lysine‑coated flasks and maintained in DMEM/F12 (1: 1) medium supplemented with 10 % fetal bovine serum at 37 °C in 5 % CO₂. Following serial passages, the adherent cell population acquired a stable, immortalized phenotype (≈105 passages).
Morphologically, ANGM‑CSS grows as a monolayer of large, polygonal to spindle‑shaped cells. By immunofluorescence, ANGM‑CSS cells are uniformly positive for CD133, nestin and vimentin, but glial fibrillary acidic protein (GFAP) is detected only in a subpopulation, indicative of heterogeneous differentiation. Karyotypically, ANGM‑CSS has a near‑tetraploid chromosome number (modal chromosome number 88-91) with the presence of recurrent unbalanced translocations t(6;14)(p12;q11.2), t(8;10)(q24.2;q21.1) and t(5;9)(q34;p21). Molecular profiling demonstrates amplification and mRNA over‑expression of MET (≈4.65‑fold) and EGFR (≈1.45‑fold) without MGMT promoter methylation or hotspot TP53/KRAS mutations.
ANGM‑CSS cells implanted subcutaneously into athymic nude mice at 5 × 10⁵ cells per injection produce palpable tumors within six weeks, with xenograft‑derived cells having the same morphology and karyotype as the parental line, indicating high tumorigenicity.
Because it preserves key GBM hallmarks-stem‑cell markers, RTK pathway activation, and in vivo tumor formation-ANGM‑CSS serves as a valuable platform for: (1) understand the molecular pathogenesis and signaling in GBM, (2) examine the efficacy of targeted anti-cancer therapeutics (e.g., MET/EGFR inhibitors) in culture, (3) perform pre‑clinical drug response studies using xenograft models, and (4) identify mechanisms of resistance to alkylating agents such as temozolomide.
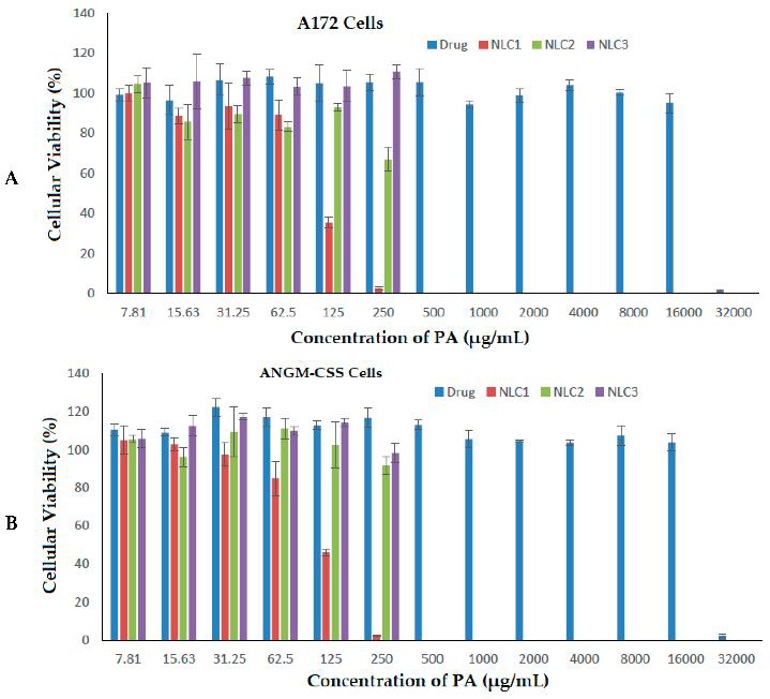
Incorporation of Perillyl Alcohol into Lipid-Based Nanocarriers Enhances the Antiproliferative Activity in Malignant Glioma Cells
Perillyl alcohol (PA) shows anti-glioma activity but suffers from poor solubility and bioavailability. Ahmed et al. aim to encapsulate PA in lipid nanocarriers (LNCs) and test their cytotoxicity against A172 and ANGM-CSS brain cancer cells.
Different doses of PA and NLC formulations were evaluated on A172 and ANGM-CSS cells over 24 h (Fig. 1A, B). Free PA oil showed high antiproliferative activity only at the highest dose (32,000 µg/mL), with IC50 values of 21,449 µg/mL and 22,037 µg/mL for A172 and ANGM-CSS cells, respectively. Loading PA into the nanocarrier delivery system (NLC1) markedly lowered IC50 to 109.7 µg/mL in A172 and 110.6 µg/mL in ANGM-CSS, outperforming the sefsol-based NLC2 (274.7 and 287.1 µg/mL) and the non-medicated NLC3 (353.2 and 335.6 µg/mL). These results suggest PA-loaded nanocarriers as potential anti-glioblastoma agents in vitro, while further studies are needed. Although NLC3 lacks drug, it still exhibits activity due to the EPR effect, and lipid encapsulation improves PA solubility by shielding it from water.
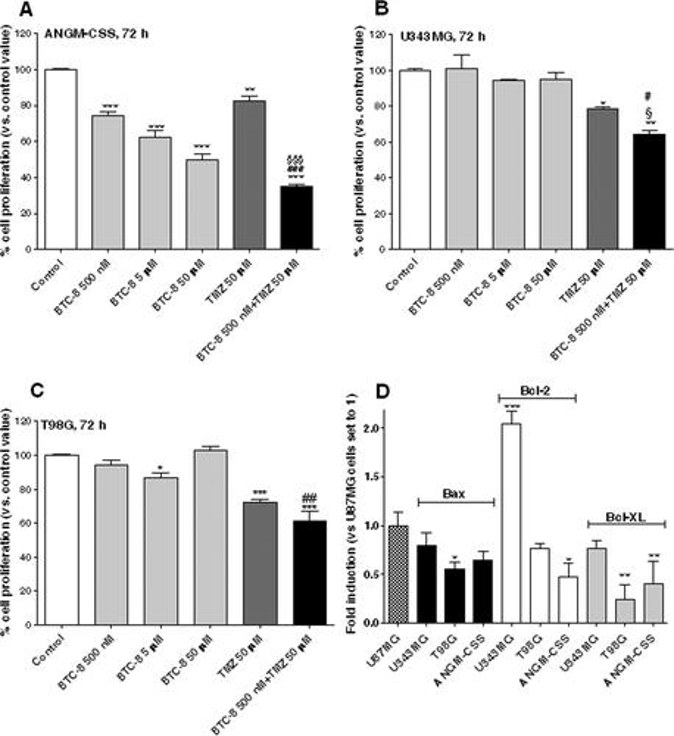
BTC-8 Differentially Affects Cell Proliferation in Distinct GBM Cell Lines
GBM resists standard chemotherapy owing to drug-efflux and apoptosis-evading glioma stem cells (GSCs). Daniele et al. tested BTC-8, a BAX activator derived from BAM-7, to determine whether direct mitochondrial apoptosis induction can eradicate bulk GBM and the therapy-resistant GSC pool.
They first tested the effects of BTC-8 on U87MG cells. Results showed BTC-8 inhibits U87MG cell proliferation and potentiates TMZ-induced antiproliferative effects. To further test the Bax activator BTC-8 across GBM heterogeneity, wild-type p53 lines U343MG and ANGM-CSS, plus mutant-p53 T98G, were examined. Figure 2A shows BTC-8 concentration-dependently suppressed ANGM-CSS growth (IC50 495.5 ± 40.8 nM), mirroring U87MG, whereas TMZ alone had only modest effect. Combined BTC-8/TMZ was significantly superior to either single agent, indicating Bax activation can sensitize TMZ-resistant cells. These data align with recent evidence that Bak, a Bax homologue, is required for TMZ-induced apoptosis, reinforcing Bax as a target to overcome TMZ resistance.
Ask a Question
Write your own review
- Adipose Tissue-Derived Stem Cells
- Human Neurons
- Mouse Probe
- Whole Chromosome Painting Probes
- Hepatic Cells
- Renal Cells
- In Vitro ADME Kits
- Tissue Microarray
- Tissue Blocks
- Tissue Sections
- FFPE Cell Pellet
- Probe
- Centromere Probes
- Telomere Probes
- Satellite Enumeration Probes
- Subtelomere Specific Probes
- Bacterial Probes
- ISH/FISH Probes
- Exosome Isolation Kit
- Human Adult Stem Cells
- Mouse Stem Cells
- iPSCs
- Mouse Embryonic Stem Cells
- iPSC Differentiation Kits
- Mesenchymal Stem Cells
- Immortalized Human Cells
- Immortalized Murine Cells
- Cell Immortalization Kit
- Adipose Cells
- Cardiac Cells
- Dermal Cells
- Epidermal Cells
- Peripheral Blood Mononuclear Cells
- Umbilical Cord Cells
- Monkey Primary Cells
- Mouse Primary Cells
- Breast Tumor Cells
- Colorectal Tumor Cells
- Esophageal Tumor Cells
- Lung Tumor Cells
- Leukemia/Lymphoma/Myeloma Cells
- Ovarian Tumor Cells
- Pancreatic Tumor Cells
- Mouse Tumor Cells