Mouse Corpus Cavernosum Smooth Muscle Cells
Cat.No.: CSC-C5320S
Species: Mouse
Source: Penis
Cell Type: Smooth Muscle Cell
- Specification
- Background
- Scientific Data
- Q & A
- Customer Review
Mouse corpus cavernosum smooth muscle cells from Creative Bioarray are isolated from the mouse penis tissue. The method we use to isolate mouse corpus cavernosum smooth muscle cells was developed based on a combination of established and our proprietary methods. The mouse corpus cavernosum smooth muscle cells are characterized by immunofluorescence with antibodies specific to α-SMA. Each vial contains 0.5x10^6 cells per ml and is delivered frozen.
Mouse Corpus Cavernosum Smooth Muscle Cells (MCCSMCs) are smooth muscle cells that are isolated from the rodent penis. They are contractile cells that make up most of the erectile tissue. Physiologically, these cells control smooth muscle tone in the spaces of Knight to facilitate erection and detumescence.
Contractile cells are also able to deconvert to synthetic phenotype. Contractile phenotype smooth muscle cells allow for both relaxation and contraction signals to be carried out. Normal contractile tone is mediated by signaling pathways. The relaxation pathway is induced by Nitric Oxide (NO)/cGMP signaling cascade. During sexual stimulation, NO from nerves or endothelial cells signals guanylate cyclase in smooth muscle cells to convert GTP to cGMP. Smooth muscle cells will then sequester calcium, resulting in muscle relaxation. On the other hand, contraction is mediated through RhoA/Rho-kinase (ROCK) and adrenergic signals. These induce smooth muscle contraction by phosphorylating myosin light chain and increasing calcium sensitivity.
Contractile cells isolated from corpus cavernosum smooth muscle cells allow for research on Erectile Dysfunction (ED). Erectile dysfunction is commonly seen in diabetic, hypertensive and metabolic syndrome patients. Current avenues of research include:
Pathophysiology: Mechanisms of how oxidative stress/free radicals or AGE's affect relaxation pathways.
Gene and Cell therapy: Efficiency of using viral vectors or stem cell secretomes to convert synthetic phenotype back to smooth muscle cells.
Drug Screening: Novel PDE5 inhibitors or alternative smooth muscle relaxing drugs.
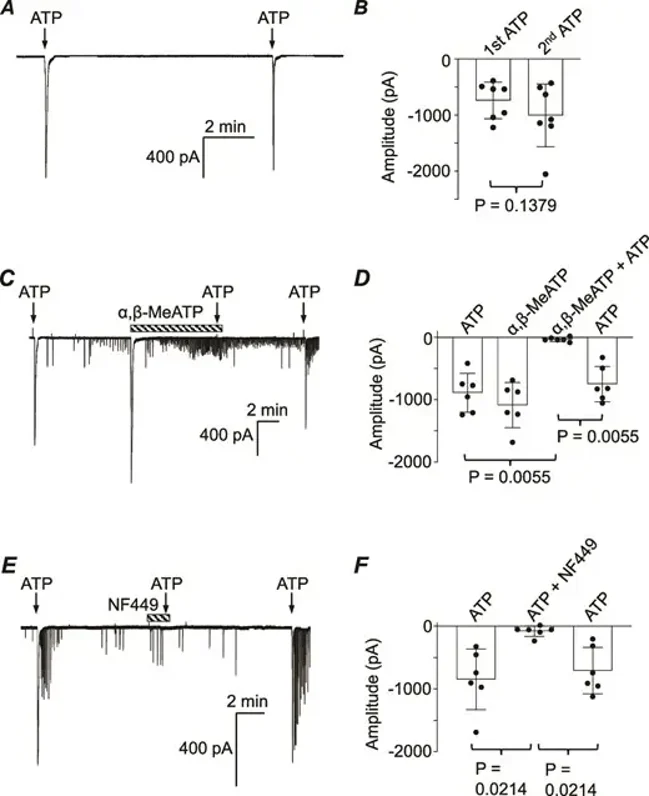
Evidence of an Excitatory Purinergic Innervation in Mouse Corpus Cavernosum Smooth Muscle
Corpus cavernosum smooth muscle (CCSM) cells are known to express P2X receptors, but their functional role in excitatory innervation is unclear. Here, Lim et al. isolated mouse CCSM myocytes and studied them using patch clamp techniques and measured isometric tension in response to electrical field stimulation (EFS).
In isolated mouse CCSM myocytes held at -60 mV, ATP (1 μm) evoked large inward currents that didn't significantly reduce in amplitude over ≥8-minute intervals (Fig. 1A). Summary data showed no significant difference between the first and second ATP applications (Fig. 1B). Next, α,β-methylene ATP (10 μm), which activates P2X1 or P2X3 receptors, was tested. It evoked a large, rapidly declining inward current. After an 8-minute washout, a second ATP application was ineffective, but a third application after another washout evoked a large current again (Fig. 1C). Summary data from six experiments confirmed that α,β-methylene ATP reversibly reduced ATP-evoked responses (Fig. 1D). NF449, a selective P2X1 receptor blocker, was then tested. ATP initially evoked a large inward current, but a second ATP application in NF449 was ineffective. After an 8-minute washout, ATP again evoked a large current. NF449 reversibly inhibited ATP-evoked currents in six experiments, confirming P2X1 receptor mediation (Fig. 1E and 1F). After the large inward currents evoked by ATP or α,β-methylene ATP declined, smaller currents resembling spontaneous transient inward currents (STICs) often appeared. Ani9 (1 μm), a TMEM16A Ca2+-activated Cl- channel blocker, completely inhibited these STICs, confirming their mediation by TMEM16A.
Ask a Question
Write your own review
- You May Also Need