Rabbit Atrial Cardiomyocytes
Cat.No.: CSC-C5189S
Species: Rabbit
Source: Heart
Cell Type: Cardiomyocyte
- Specification
- Background
- Scientific Data
- Q & A
- Customer Review
Rabbit atrial cardiomyocytes from Creative Bioarray are isolated from the rabbit heart tissue. The method we use to isolate rabbit atrial cardiomyocytes was developed based on a combination of established and our proprietary methods. The rabbit atrial cardiomyocytes are characterized by immunofluorescence with antibodies specific to myosin heavy chain. Each vial contains 0.5x10^6 cells per ml and is delivered frozen.
The Rabbit Atrial Cardiomyocyte (RAC) cell line is a continuous, non-transformed cell line derived from the atrial tissue of adult New Zealand White rabbits. It was originally established to provide a stable and reproducible in vitro model for studying atrial-specific cardiac physiology, pharmacology, and pathophysiology, addressing the limitations of primary adult cardiomyocytes which are terminally differentiated and difficult to maintain in long-term culture.
While exhibiting an epithelial-like morphology when cultured, RAC cells maintain many of the key defining properties of cardiac myocytes. RAC cells express known proteins specific to atrial myocytes including atrial natriuretic peptide (ANP), and maintain functional ion channels. Because of this, they have been used as an in vitro tool to study atrial-specific electrophysiology such as the ultra-rapid delayed rectifier potassium current (IKur), which is currently being targeted for the development of atrial-selective antiarrhythmic drugs. RAC cells have also been used to study atrial stretch, hormonal stimulation, and pro-fibrotic signaling.
The most common use for this cell line has been basic science experimentation into atrial fibrillation pathophysiology and atrial remodeling as well as screening new antiarrhythmic drug candidates. As these cells do not contract like their primary counterparts, they lack some myocardial specificity. However, they can be genetically manipulated and provide a means to culture many more cells than can be feasibly harvested from animals for high throughput assays.
Acute Effects of Axial Stretch on Force and CaT in Atrial Cardiomyocytes
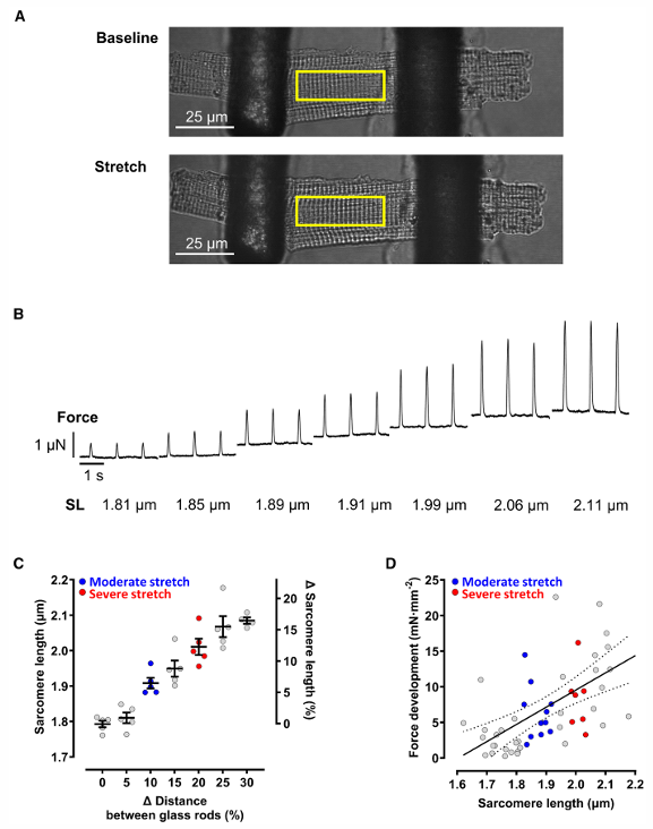
Mechanical stretch of the myocardium is proarrhythmic and alters cellular Ca2+ handling, potentially involving cation nonselective mechano- sensitive ion channels. Fu's team aimed to assess the presence and mechanisms of stretch- induced increase in Ca2+- spark rate (SiS) in isolated atrial cardiomyocytes.
Atrial cardiomyocytes were stretched axially using glass rods while paced at 2 Hz (Fig. 1A). Stretch increased diastolic sarcomere length (SL) from 1.79±0.01 μm (nonstretched) to 1.91±0.02 μm (moderate stretch, ~6%) and 2.01±0.02 μm (severe stretch, ~12%), with maximum individual SL reaching ~2.2 μm (Fig. 1B, C)-within the physiological diastolic range. Active force increased with SL (Fig. 1D), confirming the Frank-Starling response without significant systolic Ca²⁺ transient (CaT) changes.
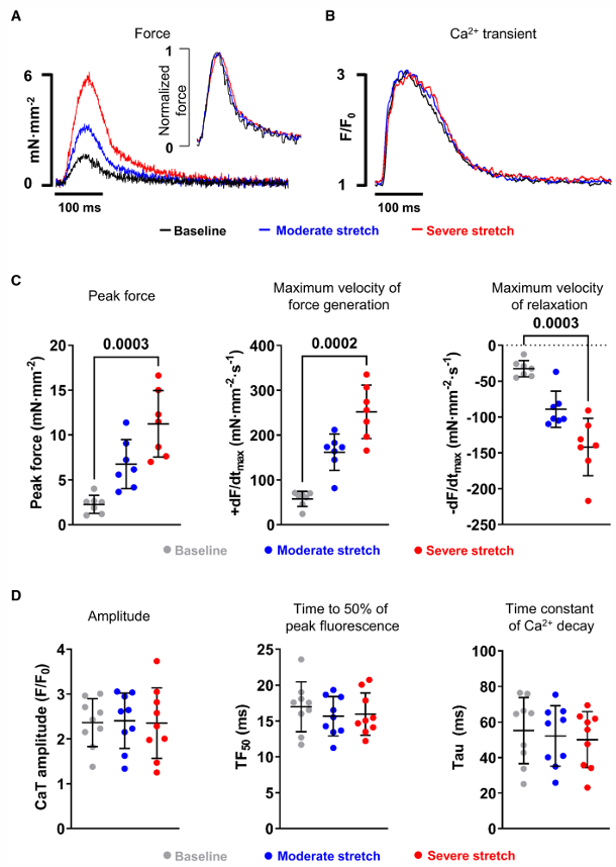
Simultaneous force and CaT measurements during 3 consecutive beats before and 20 seconds after stretch onset showed stretch-dependent increases in peak force, maximum velocity of force generation, and relaxation (Fig. 2A, C). Time-to-peak force and time from peak to 50% relaxation were unchanged. Superimposed CaT traces showed no significant changes in amplitude, time to 50% peak fluorescence, or decay time constant (Fig. 2D), confirming that the Frank-Starling response occurs without altered systolic Ca²⁺ handling.
Ask a Question
Write your own review

