SUP-T11
Cat.No.: CSC-C0605
Species: Homo sapiens (Human)
Source: Bone Marrow
Morphology: small, polymorph cells growing singly in suspension
Culture Properties: suspension
- Specification
- Background
- Scientific Data
- Q & A
- Customer Review
Immunology: CD2 +, CD3 +, CD4 -, CD5 +, CD6 +, CD7 +, CD8 -, CD13 -, CD19 -, CD20 -, CD34 -, HLA-DR -
Viruses: PCR: EBV -, HBV -, HCV -, HIV -, HTLV-I/II -, SMRV -
The SUP-T11 cell line is a highly characterized human T-lymphoblastoid line derived from a patient with T-cell lymphoblastic lymphoma. Recognized for its unique phenotypic profile and robust growth characteristics, SUP-T11 serves as a critical in vitro model for studying T-cell receptor (TCR) signaling, leukemogenesis, and the molecular mechanisms of viral entry and replication.
- High CD4+ and CXCR4 Expression: SUP-T11 cells are naturally high expressors of both CD4 and the CXCR4 coreceptor. This specific surface profile makes them an exceptional substrate for studying HIV-1 infection, viral fusion assays, and the development of entry inhibitors, offering superior physiological relevance over engineered cell lines.
- TCR Rearrangement & Signaling: As a T-lineage model, SUP-T11 retains key elements of the T-cell signaling machinery. This makes it a valuable platform for investigating V(D)J recombination and chromosomal translocations common in hematologic malignancies, as well as for validating novel CAR-T cell constructs.
- Exceptional Growth and Scalability: These cells exhibit consistent, rapid proliferation in standard RPMI-1640 media. Their stable suspension growth facilitates easy scaling for High-Throughput Screening (HTS) of anti-leukemic compounds and large-scale viral production.
- Proven Genetic Stability: Our SUP-T11 stocks are rigorously authenticated via STR profiling and confirmed to be free of mycoplasma. Their predictable genetic background ensures high reproducibility in gene editing studies using CRISPR/Cas9 or RNA interference (RNAi).
By incorporating the SUP-T11 cell line into your oncology or virology pipeline, you utilize a reliable, well-documented human model designed to provide the clarity and consistency required for high-impact discovery.
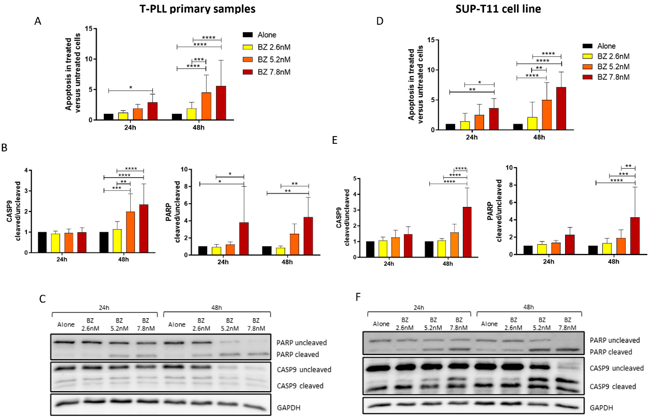
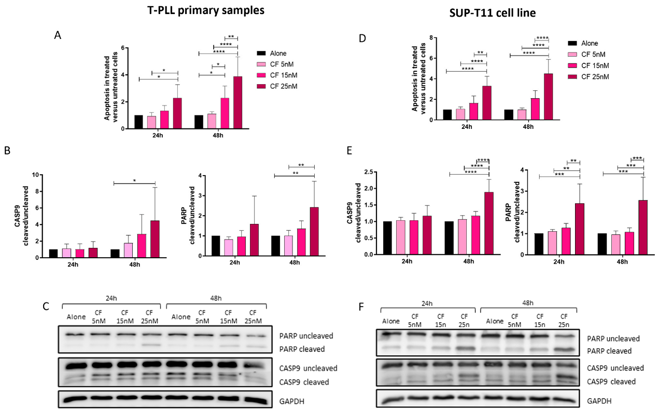
Proteasome Inhibitors Induce Apoptosis in Ex Vivo Cells of T-Cell Prolymphocytic Leukemia (T-PLL)
Finding an effective treatment for T-PLL patients remains a significant challenge. Alemtuzumab, currently the gold standard, is insufficient in managing the aggressiveness of the disease in the long term. Consequently, numerous efforts are underway to address this unmet clinical need.
The rarity of the disease limits the ability to conduct robust clinical trials, making in silico, ex vivo, and in vivo drug screenings essential for designing new therapeutic strategies for T-PLL. We conducted a drug repurposing analysis based on T-PLL gene expression data and identified proteasome inhibitors (PIs) as a promising new class of compounds capable of reversing the T-PLL phenotype. Treatment of ex vivo T-PLL cells and the SUP-T11 cell line with Bortezomib and Carfilzomib, two PI compounds, supported this hypothesis by demonstrating increased apoptosis in leukemic cells. Taken together, these findings open new avenues for investigating the molecular mechanisms underlying the efficacy of PI in T-PLL and expand the spectrum of potential therapeutic strategies for this highly aggressive disease.
Ask a Question
Write your own review
- You May Also Need
- Adipose Tissue-Derived Stem Cells
- Human Neurons
- Mouse Probe
- Whole Chromosome Painting Probes
- Hepatic Cells
- Renal Cells
- In Vitro ADME Kits
- Tissue Microarray
- Tissue Blocks
- Tissue Sections
- FFPE Cell Pellet
- Probe
- Centromere Probes
- Telomere Probes
- Satellite Enumeration Probes
- Subtelomere Specific Probes
- Bacterial Probes
- ISH/FISH Probes
- Exosome Isolation Kit
- Human Adult Stem Cells
- Mouse Stem Cells
- iPSCs
- Mouse Embryonic Stem Cells
- iPSC Differentiation Kits
- Mesenchymal Stem Cells
- Immortalized Human Cells
- Immortalized Murine Cells
- Cell Immortalization Kit
- Adipose Cells
- Cardiac Cells
- Dermal Cells
- Epidermal Cells
- Peripheral Blood Mononuclear Cells
- Umbilical Cord Cells
- Monkey Primary Cells
- Mouse Primary Cells
- Breast Tumor Cells
- Colorectal Tumor Cells
- Esophageal Tumor Cells
- Lung Tumor Cells
- Leukemia/Lymphoma/Myeloma Cells
- Ovarian Tumor Cells
- Pancreatic Tumor Cells
- Mouse Tumor Cells