- Specification
- Background
- Scientific Data
- Q & A
- Customer Review
DI TNC1 is an immortalized rat astrocyte cell line established from primary astrocytes isolated from neonatal rat brain and then transformed with SV40 large T antigen. It preserves most astrocytic phenotypic and functional properties and has been frequently used as an in vitro astrocyte model system to study glial biology and CNS pathophysiology.
DI TNC1 cells are stellate/polygonal shaped resembling astrocytes and form adherent monolayers when cultured. These cells express GFAP, and have many functional properties of astrocytes including extracellular ion regulation and regulation of neuronal metabolism. DI TNC1 cells, unlike primary astrocytes, have better proliferation characteristics and are more easily able to be reproduced.
DI TNC1 cells have been used in a variety of experiments modeling inflammation, oxidative stress and astrocyte-mediated neurotoxicity. This includes studying mechanisms of excitotoxicity, responses to cytokines, and responses to stimuli associated with neurodegenerative diseases. DI TNC1 cells have also been used for neurotoxicity assays and mechanistic evaluations of drug candidates to assess CNS safety.
Lysophosphatidylcholine is not Toxic to DI TNC1 Astrocytes but Exerts a Pro-Inflammatory Effect at the Signaling Level
Astrocytes play an important role in the regulation of the inflammatory response in the CNS, e.g., in demyelinating diseases. Since the chemokine CXCL1 is known to be secreted by astrocytes and to have a pro-inflammatory effect on immune cells in the CNS, Turniak-Kusy et al. verified the effect of testosterone on its secretion in vitro (in the astrocytic cell line DI TNC1).
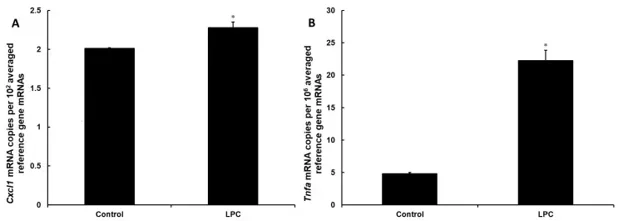
Since incubation with the known in vivo inflammation stimulant lysophosphatidycholine (LPC) can change the composition of cell plasma membrane, leading to cytotoxicity, they verified the cytotoxic effect of LPC on DI TNC1 astrocytes. Cells were treated with 0-200 µg/mL LPC for 24 h and membrane integrity assessed by Hoechst 33342 (permeable) and propidium iodide (impermeable) staining. At all concentrations, <1% of nuclei were propidium iodide-positive, indicating preserved membrane integrity. They then tested pro-inflammatory effects of 150 µg/mL LPC. At the mRNA level, Tnfa expression increased 360% (Fig. 1A), while Cxcl1 was induced by 13% (Fig. 1B), providing mechanistic basis for enhanced chemokine secretion following LPC treatment.
Ask a Question
Write your own review
- Adipose Tissue-Derived Stem Cells
- Human Neurons
- Mouse Probe
- Whole Chromosome Painting Probes
- Hepatic Cells
- Renal Cells
- In Vitro ADME Kits
- Tissue Microarray
- Tissue Blocks
- Tissue Sections
- FFPE Cell Pellet
- Probe
- Centromere Probes
- Telomere Probes
- Satellite Enumeration Probes
- Subtelomere Specific Probes
- Bacterial Probes
- ISH/FISH Probes
- Exosome Isolation Kit
- Human Adult Stem Cells
- Mouse Stem Cells
- iPSCs
- Mouse Embryonic Stem Cells
- iPSC Differentiation Kits
- Mesenchymal Stem Cells
- Immortalized Human Cells
- Immortalized Murine Cells
- Cell Immortalization Kit
- Adipose Cells
- Cardiac Cells
- Dermal Cells
- Epidermal Cells
- Peripheral Blood Mononuclear Cells
- Umbilical Cord Cells
- Monkey Primary Cells
- Mouse Primary Cells
- Breast Tumor Cells
- Colorectal Tumor Cells
- Esophageal Tumor Cells
- Lung Tumor Cells
- Leukemia/Lymphoma/Myeloma Cells
- Ovarian Tumor Cells
- Pancreatic Tumor Cells
- Mouse Tumor Cells