Human iPSC-derived Osteoblasts
- Specification
- Background
- Scientific Data
- Q & A
- Customer Review
Human iPSC-derived Osteoblasts (hiPSC-OBs) are osteoblasts that are created by the directed differentiation of human induced Pluripotent Stem Cells (hiPSCs). iPSCs are stem cells that have been reprogrammed from adult somatic cells, such as dermal fibroblasts or peripheral blood cells. The differentiation process recapitulates osteogenesis in vivo. This begins with the induction of hiPSCs to become mesenchymal stem cells (MSCs) by activating BMP4 or TGF-β signaling and osteogenic commitment by stimulating the Wnt/β-catenin pathway and adding osteogenic supplements (ascorbic acid, β-glycerophosphate, dexamethasone), which induces osteoblast maturation. As they mature, they aggregate into mineralized nodules-hallmarks of osteoblastic function-while maintaining a stable karyotype and osteogenic lineage commitment.
Functionally, hiPSC-OBs possess core osteoblastic properties: extracellular matrix (ECM) synthesis (collagen type I, osteocalcin) and matrix mineralization, critical for bone formation. These traits make them pivotal in multiple fields. In basic science, hiPSC-OBs allow for the recapitulation of human osteogenesis in vitro, allowing for the study of mechanisms and signals of osteogenic lineage specification and mineralization that cannot be gleaned in animal models. In drug discovery, they can be used as a platform for screening of osteoprotective small molecules and chemicals in a human-relevant context, rather than in animal models that are often used as first line screening methods. They are also important in the field of regenerative medicine; preclinical models have demonstrated their usefulness in bone tissue engineering-seeding hiPSC-OBs on scaffolds allowed for the repair of bone defects in cases of fracture and craniofacial abnormalities.
Cellular Behavior of hiPSC-Derived Osteoblasts on a Bone-Mimetic Collagen Scaffold
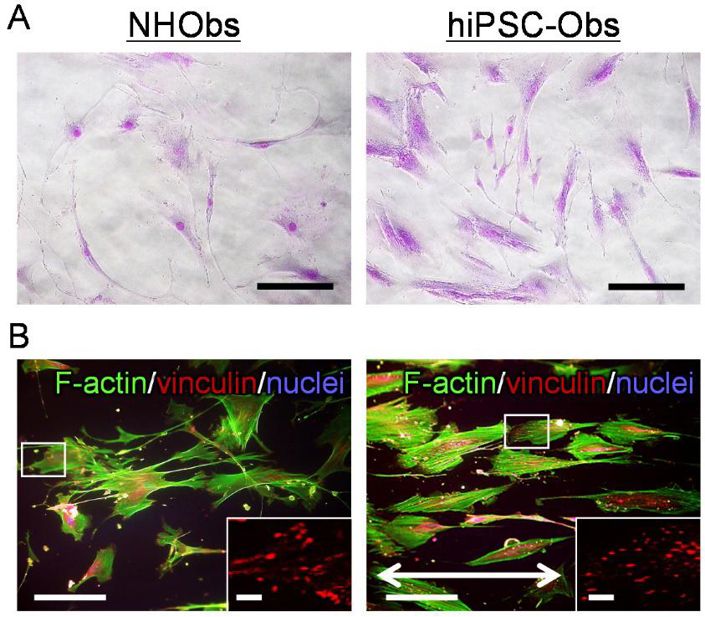
hiPSC-derived osteoblasts (hiPSC-Obs) have shown preferential alignment and organization in bone-mimetic collagen scaffolds, highlighting their role in unidirectional cellular arrangement. However, the mechanisms underlying their oriented tissue construction properties remain unclear. Matsugaki et al. aim to characterize the properties of hiPSC-Obs and their focal adhesions (FAs) to understand their role in anisotropic bone microstructural regeneration.
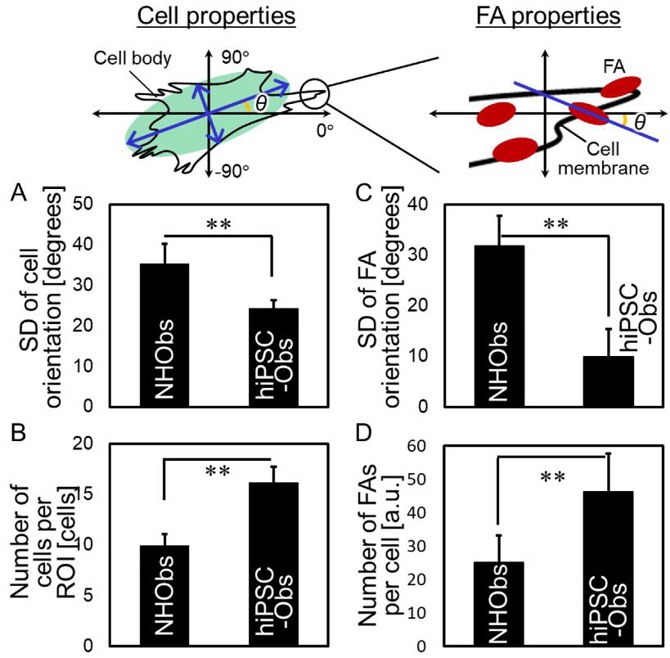
The cellular properties and focal adhesions (FAs) of hiPSC-Obs were characterized by analyzing their behavior on a bone-mimetic collagen scaffold (Fig. 1 and Fig. 2). On polystyrene plates, both hiPSC-Obs and NHObs exhibited non-uniform shapes and random orientations (Fig. 1A). On the collagen scaffold, both types of osteoblasts showed elongated bodies and aligned predominantly along the collagen fibers (Fig.1B). The collagen scaffold promoted preferential cellular orientation. Notably, hiPSC-Obs showed better responsiveness than NHObs, with decreased deviation of the cellular orientation angle (Fig. 1B and Fig. 2A) and increased cell proliferation (Fig. 2B). Additionally, hiPSC-Obs exhibited an ordered array of FAs parallel to the cell orientation axis (Fig. 1B and Fig. 2C) and an increased number of FAs (Fig. 1B and Fig. 2D).
Ask a Question
Write your own review
- Adipose Tissue-Derived Stem Cells
- Human Neurons
- Mouse Probe
- Whole Chromosome Painting Probes
- Hepatic Cells
- Renal Cells
- In Vitro ADME Kits
- Tissue Microarray
- Tissue Blocks
- Tissue Sections
- FFPE Cell Pellet
- Probe
- Centromere Probes
- Telomere Probes
- Satellite Enumeration Probes
- Subtelomere Specific Probes
- Bacterial Probes
- ISH/FISH Probes
- Exosome Isolation Kit
- Human Adult Stem Cells
- Mouse Stem Cells
- iPSCs
- Mouse Embryonic Stem Cells
- iPSC Differentiation Kits
- Mesenchymal Stem Cells
- Immortalized Human Cells
- Immortalized Murine Cells
- Cell Immortalization Kit
- Adipose Cells
- Cardiac Cells
- Dermal Cells
- Epidermal Cells
- Peripheral Blood Mononuclear Cells
- Umbilical Cord Cells
- Monkey Primary Cells
- Mouse Primary Cells
- Breast Tumor Cells
- Colorectal Tumor Cells
- Esophageal Tumor Cells
- Lung Tumor Cells
- Leukemia/Lymphoma/Myeloma Cells
- Ovarian Tumor Cells
- Pancreatic Tumor Cells
- Mouse Tumor Cells